Gene to GMP.
Faster, Higher, Smarter
Gene to GMP.
Faster, Higher, Smarter
Syngene Recombinant Protein Services
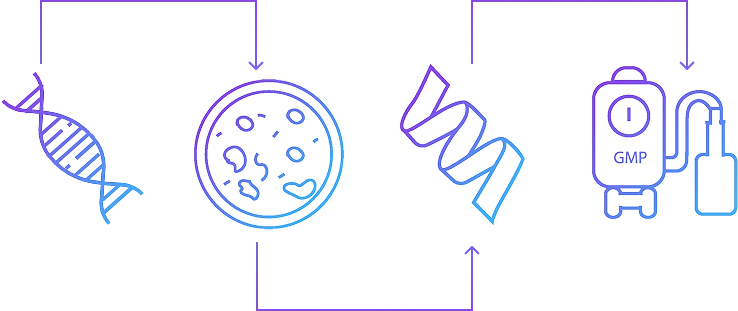
Comprehensive Recombinant Protein Services, From Gene to GMP
Syngene offers end-to-end solutions for recombinant protein production — from gene cloning and expression optimization across diverse host systems to large-scale manufacturing for pre-clinical and clinical development.
At the core is our SynWeave™ rapid protein production platform, which boosts efficiency, shortens timelines, and delivers consistently high yields.
Our services span a broad spectrum of protein types, including monoclonal antibodies (mAbs), biosimilars, bispecific antibodies, and other biologics — helping clients bring novel therapies to market faster, with confidence.
SynWeave™ Advantage
SynWeave™ integrates cutting-edge ExcellGene transposon technology with Syngene’s process expertise to deliver titers up to 12 g/L — reducing development timelines by as much as 10 weeks.
10 weeks faster to
pre-clinical/clinical
7–12 g/L titers
achieved
Global GMP sites in
India & US
End-to-end services
from gene to GMP
Capabilities at a Glance
End-to-End Recombinant Protein Services

Integrated Services

Rapid Protein Production Platform (SynWeave™)
Versatile Host Systems
Broad Range of Protein Types
Manufacturing Capabilities

Focus on Efficiency

Applications
SynWeave™ Advantage
SynWeave™:
Redefining Protein Production
SynWeave™ leverages state-of-the-art cell line and transposon-based technologies, integrates ExcellGene’s advanced transposon-based cell line technology with Syngene’s process development expertise. The result: faster, more reliable recombinant protein production with superior manufacturability and efficiency.

Accelerated Development

Increased Productivity
