Toxicology has always been a decisive discipline in drug development, and today digital pathology is playing an increasingly important role in shaping how safety is assessed. It defines the boundary between promising pharmacology and acceptable clinical risk. Historically, safety evaluation relied heavily on animal studies supported by standard in vitro assays and exposure measurements. While these approaches remain essential, current developments in toxicology—supported by computational toxicology, predictive toxicology, and new approach methodologies—are reshaping how risk is understood, predicted, and managed.
Today’s safety science is becoming more predictive, more mechanistic, and more integrated. Computational toxicology, New Approach Methodologies (NAMs), pathway-based in vitro systems, high-content imaging, and digital pathology are expanding the scientific toolkit available to drug developers. At the same time, regulatory thinking is evolving toward weight-of-evidence (WoE) frameworks that integrate diverse data streams into coherent safety narratives.
The transition from descriptive to predictive toxicology
Traditional toxicology has largely been descriptive, establishing no‑observed‑adverse‑effect levels (NOAELs) from animal studies and extrapolating to first‑in‑human (FIH) dosing with safety factors. Although effective, this model has inherent limitations. Animal systems may not fully replicate human biology, mechanistic interpretation is sometimes limited, and unexpected toxicities can still emerge during clinical development.
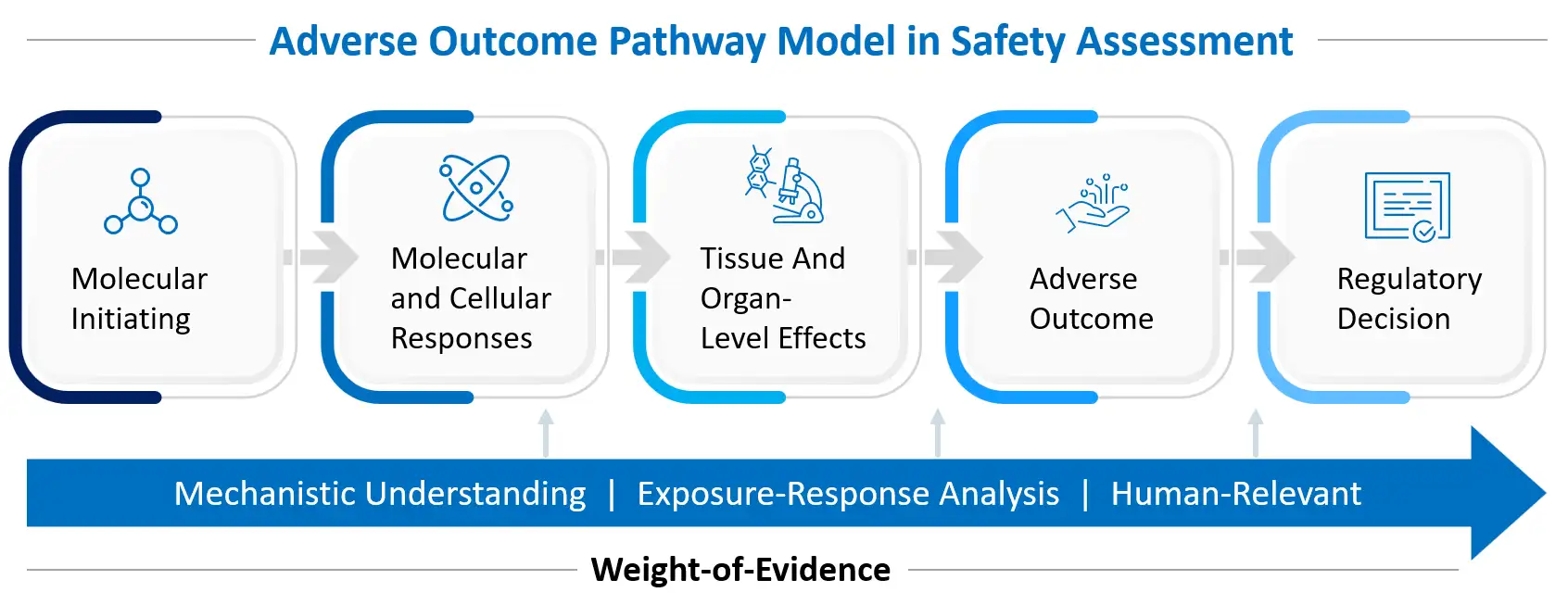
Modern toxicology is moving towards predictive, seeking early molecular and cellular perturbations that precede overt toxicity. This shift is grounded in the concept of adverse outcome pathways (AOPs), which map the progression from a molecular initiating event to tissue and organism-level effects to support decision making. The structured progression from early molecular perturbation to organ-level toxicity and ultimately regulatory decision-making is illustrated in the adverse outcome pathway model shown below. By understanding toxicity at the pathway level, scientists can better anticipate risk, quantify margins, and design mitigation strategies before clinical exposure occurs.
Mechanism-based in vitro systems, new approach methodologies, and pathway-level toxicology
Mechanism-driven safety assessment has become central to current developments in toxicology. Rather than relying solely on general cytotoxicity assays, researchers now deploy targeted in vitro systems to probe specific biological pathways.
These systems evaluate processes such as mitochondrial impairment, oxidative stress signaling, unfolded protein response, inflammatory activation, DNA damage response, and ion channel disruption. For example, mitochondrial stress assays can reveal early liabilities linked to hepatotoxicity, while cardiomyocyte-based models can detect electrophysiological disturbances associated with arrhythmogenic risk.
High-content imaging has significantly strengthened this approach. Using automated microscopy combined with advanced image analysis, scientists can quantify multiple cellular features simultaneously—nuclear morphology, organelle integrity, membrane permeability, cytoskeletal changes, and biomarker localization. This multi-parametric analysis generates rich datasets that improve sensitivity and specificity compared with single-endpoint assays.
Mechanistic data also support exposure–response interpretation. When in vitro pathway activation is linked to pharmacokinetic modelling, teams can better estimate the safety margin between therapeutic exposure and toxicity thresholds.
Computational toxicology and artificial intelligence in predictive safety
Computational toxicology has rapidly evolved into a powerful complement to experimental science. Advances in machine learning, cheminformatics, and big data analytics now enable the integration of chemical structure information with historical toxicology datasets.
Quantitative structure–activity relationship (QSAR) models can identify structural motifs associated with specific toxicities. In parallel, machine-learning algorithms trained on large, curated datasets can predict organ-specific risk, off-target receptor binding, and metabolic liabilities.
These tools are particularly valuable during early discovery and lead optimization. By predicting potential liabilities before synthesis or in vivo testing, computational models help prioritize candidates with improved safety profiles. This approach strengthens predictive toxicology strategies andreduces attrition, conserves resources, and shortens development timelines.
Artificial intelligence is also being applied to interpret complex biological datasets generated by high-content imaging, transcriptomics, proteomics, and, metabolomics. Pattern recognition algorithms can detect subtle phenotypic signatures associated with emerging toxicity, enhancing predictive accuracy.
Importantly, computational models are most effective when used within an integrated framework. Predictions must be validated experimentally, and any discrepancies should inform model refinement. This cycle strengthens both the computational and experimental components of safety assessment and supports regulatory acceptance of in silico approaches.
Microphysiological systems and organ-on-chip technologies
One of the most promising areas in human-relevant toxicology is the development of microphysiological systems (MPS), often described as organ-on-chip technologies. These engineered platforms recreate aspects of tissue microarchitecture, perfusion, and cell–cell interactions under controlled conditions.
Unlike traditional two-dimensional cell cultures, MPS incorporate dynamic flow, extracellular matrix components, and multiple cell types. This complexity allows better modelling of physiological responses, including metabolism, barrier function, and immune interactions.
Liver-on-chip models allow evaluation of drug-induced liver injury (DILI) under physiologically relevant conditions. Cardiac microtissue systems support assessment of contractility and electrophysiological stability. Kidney and blood-brain barrier chips provide insights into organ-specific transport and injury mechanisms.
When integrated with pharmacokinetic modeling, microphysiological data can inform systemic exposure predictions and refine margin-of-safety calculations. Although these technologies are still evolving, they represent a critical advance in bridging preclinical models and human trials, reducing translational uncertainty, particularly when combined with new approach methodologies.
Integration of toxicokinetics and exposure science
Safety cannot be interpreted without understanding exposure. Toxicokinetics (TK) and pharmacokinetics (PK) remain foundational to toxicology. Modern analytical platforms, particularly advanced liquid chromatography–mass spectrometry (LC–MS) systems, now offer highly sensitive and specific quantification of parent compounds and metabolites. These technologies support precise exposure–toxicity correlations. By linking systemic and tissue concentrations with observed biological effects, teams can derive more accurate no observed adverse effect levels (NOAELs) and safety margins.
Metabolite identification is also increasingly critical, as some toxicities arise not from the parent compound but from reactive or unstable metabolites. High-resolution mass spectrometry and metabolomic profiling help detect these species early, enabling chemical modification strategies to reduce risk.
Integration of PK, TK, and mechanistic in vitro data strengthens translational modeling. Physiologically based pharmacokinetic (PBPK) models, informed by experimental parameters, support prediction of human exposure scenarios and dose selection strategies. When combined with exposure, PBPK models also allow simulation of vulnerable populations (e.g., pediatrics, elderly, or those with impaired organ function), enhancing risk assessment relevance.
Digital pathology and quantitative tissue analysis
Histopathology remains a cornerstone of safety evaluation, but it is also undergoing significant transformation. Traditional microscopy interpretation, while expert-driven, can involve subjectivity and inter-observer variability. Digital pathology platforms now allow high-resolution slide scanning, algorithm-driven quantification, and standardized reporting. Image analysis software can measure tissue architecture, inflammatory infiltration, fibrosis markers, and cellular degeneration in a reproducible and quantitative manner.
This digital transformation enhances reproducibility and enables integration of histological data with molecular and exposure information. Quantitative tissue metrics not only strengthen statistical analysis but also improve regulatory communication and cross-study comparability. Furthermore, advances in artificial intelligence and machine learning algorithms are increasingly being applied to digital pathology, allowing automated detection of subtle morphological changes and predictive modeling of toxicity outcomes. This positions digital pathology as a key enabler of predictive toxicology and integrated safety assessment.
Regulatory evolution and weight-of-evidence frameworks
Regulatory agencies worldwide are increasingly supportive of integrated, science-based safety strategies. Rather than focusing on isolated study outcomes, regulators emphasize comprehensive weight-of-evidence (WoE) evaluation. In this model, in silico predictions, mechanism-based in vitro findings, in vivo toxicology data, and exposure analysis are considered collectively. Concordance across these streams enhances confidence in risk assessment.
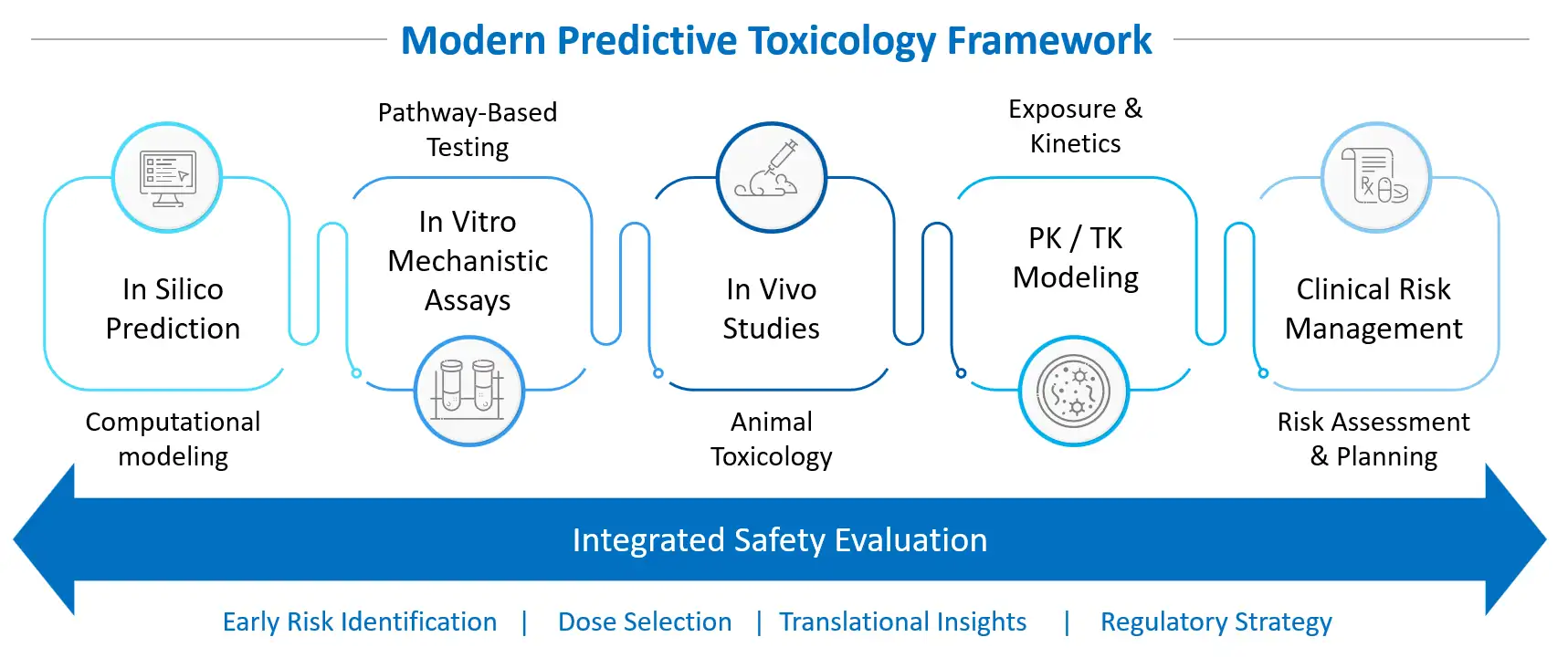
The growing recognition of new approach methodologies (NAMs) reflects this evolution. Although animal studies remain important for systemic safety assessment, there is a clear regulatory interest in scientifically validated alternatives that improve human relevance and reduce unnecessary animal use. This convergence of in silico prediction, mechanism-based in vitro assays, targeted in vivo confirmation, and PK/TK modeling is illustrated in the modern predictive toxicology framework below.
Mechanistic justification is becoming increasingly critical in regulatory submissions. Sponsors are expected not only to describe observed findings but also to explain the underlying biological mechanisms and their relevance to humans. This shift aligns with regulatory guidance from agencies such as the U.S. FDA, EMA, and OECD, which encourage mechanistic data integration to strengthen translational confidence.
Furthermore, weight-of-evidence frameworks are being supported by structured decision-making tools, such as adverse outcome pathways (AOPs), which provide mechanistic context for toxicity endpoints. Integration of AOPs with NAMs and PBPK modeling enhances regulatory acceptance and facilitates transparent communication of risk.
Importance of integrated, co-localized R&D ecosystems
Technological innovation alone does not guarantee translational success. Organizational structure plays a crucial role. When discovery, DMPK, safety assessment, and early clinical enablement teams operate within a co-localized and integrated R&D environment, scientific communication improves significantly.
Real-time, bi-directional data exchange allows toxicological signals to be interpreted in the context of pharmacology and exposure. Hypotheses can be tested rapidly, and experimental strategies can be adjusted without delay. This integration reduces siloed decision-making and supports coherent end to end development strategies.
For example, if mechanistic in vitro assays indicate mitochondrial stress at certain exposure levels, DMPK teams can model systemic concentrations to determine clinical relevance. Clinical teams can simultaneously design monitoring strategies for potential biomarkers. Such coordination improves both efficiency and safety.
Organizations such as Syngene, which operate with integrated discovery-to-early development capabilities, demonstrate how co-localized expertise can support smoother data flow and stronger translational alignment. The broader message for the industry is that integration—both scientific and operational—is increasingly critical to managing complexity in modern pipelines.
Implications for complex and emerging modalities
Current toxicology developments are particularly relevant for complex therapeutic modalities, including biologics, bispecific antibodies, antibody-drug conjugates, gene therapies, and cell-based therapies. These products often involve unique mechanisms of action, immune modulation, or targeted delivery systems.
Mechanistic understanding is essential for evaluating on-target and off-target effects. High-content imaging, immune pathway assays, and microphysiological systems can provide early insight into cytokine release potential, organ-specific accumulation, or unintended cellular interactions.
Similarly, computational modeling supports the prediction of immunogenicity risk and off-target engagement. As pipelines diversify, integrated and predictive safety frameworks become indispensable.
Toward a more strategic role for toxicology
The cumulative effect of these current toxicology developments is a shift in how safety science is positioned within drug development. Toxicology is no longer a downstream checkpoint; it is increasingly a strategic partner in decision-making.
By combining computational prediction, pathway-based evaluation, advanced analytics, and integrated organizational models, safety teams can identify risk earlier and influence molecule design proactively. This reduces late-stage failures and supports more confident progression into clinical trials.
The future of toxicology lies in integration—across data types, technologies, disciplines, and organizational structures. As predictive models improve and human-relevant systems gain validation, safety science will continue to evolve from descriptive observation toward quantitative, mechanism-informed risk assessment.
For drug development teams, embracing these advances is not optional. It is essential for reducing uncertainty, optimizing resources, and accelerating the journey from promising science to meaningful patient outcomes.