Topical drug development looks simple from the outside: patients apply a cream, gel, or ointment and expect local relief. However, as the recent Syngene webinar “Targeted Dermal Delivery: Integrating ADME Science in Topical Drug Development – Syngene International Ltd” shows, there is a sophisticated science engine behind every successful topical product. ADME behaviour in the skin, target engagement, formulation choices, and careful preclinical research all decide whether a dermal delivery program will progress or fail. Tirbanibulin is a strong example of how this science translates to patient benefit.
This webinar, part of Syngene’s DMPK Unravelled series, brought together three internal experts. The session was moderated by Dr Vishwottam Kandikere, Assistant Vice President and Head of DMPK at Syngene International Ltd. Miquel Salva, Executive Director, DMPK, shared a detailed view of topical development and target engagement using the example of tirbanibulin for actinic keratosis. Ashwani Gaur, Assistant Director, DMPK, SSSL, Hyderabad, focused on preclinical research, formulation development, and practical challenges in dermal delivery.
Rather than simply reporting the event, this article uses their core ideas to present an independent overview of how ADME science supports targeted dermal delivery, while referring back to the webinar as a key learning resource.
Tirbanibulin, target engagement, and dermal delivery science
A key point emphasized by Dr. Miquel Salva, DMPK expert and Executive Director at Synvent, Syngene’ s integrated drug discovery platform, is that skin penetration alone is not enough. For any topical drug, crossing the stratum corneum is only the first step. True success depends on whether the drug reaches its pharmacological targets in the epidermis or dermis at concentrations and for a duration that are sufficient to trigger the desired biological response.
Tirbanibulin, a tubulin inhibitor approved for actinic keratosis, is a good example. Actinic keratosis is driven by abnormal proliferation of keratinocytes that resist apoptosis. Tirbanibulin binds at the colchicine site on tubulin and also interferes with a Src kinase–related pathway. Together these actions lead to cell cycle arrest and apoptotic cell death in the diseased cells. For this mechanism to work in real patients, the tirbanibulin ointment must deliver enough drug into the epidermis while keeping systemic levels very low.
This is different from classical systemic drug development, where plasma pharmacokinetics often act as a surrogate for tissue exposure and efficacy. In topical therapy, plasma levels mainly help to monitor safety. Efficacy is tied to local dermal exposure, which must be studied through appropriate models, dermal delivery experiments, and clinical readouts.
Building a translational chain for dermal ADME
Modern dermal delivery uses a stepwise set of tools that link discovery to the clinic. At the earliest stage, in silico models and high-throughput assays such as skin PAMPA enable rapid ranking of analogues based on their ability to cross a synthetic lipid layer that mimics the stratum corneum. These models are useful for identifying trends but do not capture the complexity of real human skin.
The next step is the use of Franz diffusion cells. In this system, human or animal skin is mounted between donor and receptor chambers. The test topical formulation is applied on the surface, and samples are taken from the receptor compartment over time. This setup preserves key barrier features and allows direct comparison between different topical formulation development options. Regulators recognize data from such models, and they form a central part of the package that supports dose selection.
Finally, maximal use systemic exposure studies in humans test the formulation under worst-case conditions. In tirbanibulin studies, 1 percent ointment was applied to fields of 25 square centimetres and 100 square centimetres in actinic keratosis patients once daily for five days. Systemic exposure increased roughly in proportion to the treated area but remained in the low nanogram-per-millilitre range and showed no accumulation over time. Combined with strong phase 3 efficacy data, these findings support tirbanibulin as a safe and effective field therapy.
Preclinical research and skin research for topical drugs
Ashwani Gaur’s perspective highlights how preclinical research in dermatology needs both creativity and discipline. Topical systems offer several advantages. They provide local treatment at the disease site, reduce systemic effects, avoid first-pass metabolism, and support many dosage forms such as creams, gels, ointments, lotions, foams, sprays, and patches. It is not surprising that the global market for such formulations is expanding steadily.
Yet dermal delivery remains challenging. The skin barrier prefers small, lipophilic, mostly unionized molecules, usually below a molecular weight of around five hundred. Many promising systemic drugs do not meet these criteria, which forces teams to rethink structures or design advanced topical formulation development strategies. Poor aqueous solubility, instability on the skin surface, and the risk of irritation or sensitization from excipients all complicate matters.
A typical development path starts with biological target identification and screening, followed by in vitro evaluation that focuses on lipophilicity, permeability, and an effective balance between penetration and potency. Cutaneous pharmacokinetics then become important. Techniques such as tape stripping, skin micro-dialysis, and biopsies help to quantify drug levels in the stratum corneum, interstitial fluid, and deeper tissues. Specific toxicology packages include skin irritation, sensitization, phototoxicity, repeated dermal toxicity, and sensory properties. As dermal delivery programs advance, work such as in vitro skin irritation tests and pharmaceutical formulation development become critical checkpoints before first-in-human dosing.
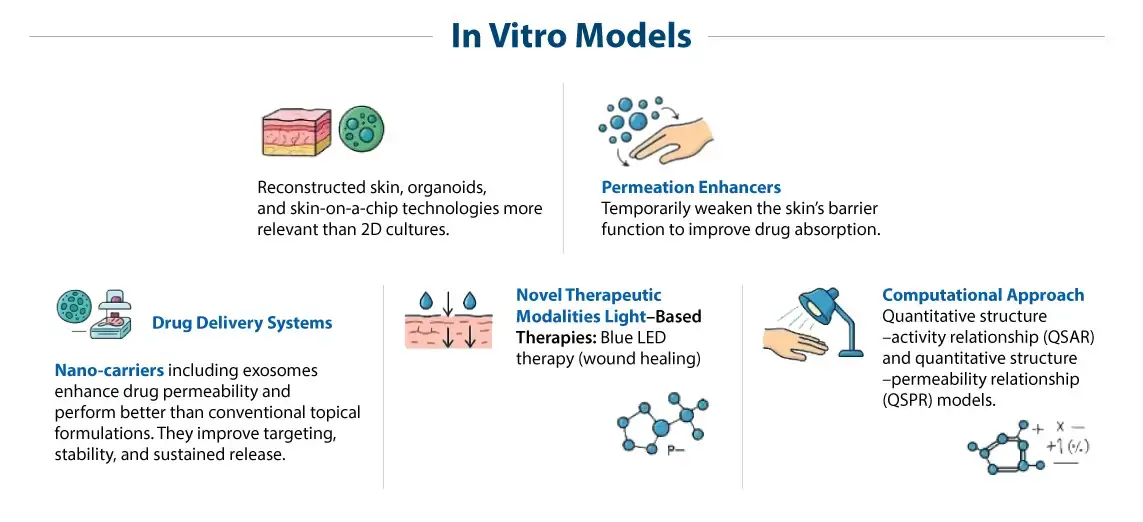
Physicochemical and formulation considerations
Formulation strategy underpins all these steps in topical drug development. Creams can be oil-in-water or water-in-oil and are versatile for many dermal applications. Gels are non-greasy and well accepted for localized treatment, though their pH and polymer system must suit both the drug and the skin. Ointments provide strong occlusion for very dry skin and highly lipophilic molecules. Lotions and sprays are suitable for large or hairy areas such as the scalp. Patient preference and disease site guide these choices as much as pure pharmacology.
Tirbanibulin demonstrates how these choices come together in practice. Its approved field-therapy ointment relies on a simple, well-tolerated vehicle that supports adequate penetration, minimal systemic exposure, and consistent patient use over a short five-day course.
Advanced topical formulation development and delivery platforms
Beyond traditional vehicles, topical formulation development is rapidly adopting nanocarriers and other advanced platforms. Liposomes, solid lipid nanoparticles, nano-emulsions, ethosomes, and cyclodextrin-based complexes can improve stability, enhance penetration, and sometimes allow sustained release. They are especially helpful when the active ingredient is poorly soluble or prone to rapid metabolism near the skin surface.
One case from Syngene’s internal dermal delivery work illustrates how formulation and route selection can rescue a promising molecule. In this project, intravenous and oral dosing caused animal mortality, with no measurable exposure after a few hours. By switching to topical administration and systematically testing multiple formulations, the team achieved high drug levels in the skin with much lower plasma concentrations. Measures such as Cmax and overall exposure dropped into a safer range, while efficacy was preserved. A key tool here was skin micro-dialysis in rats, which allowed direct measurement of free drug in the dermal interstitial fluid and helped to differentiate between total exposure and pharmacologically active levels.
Looking ahead: integrating ADME science into dermal delivery
When viewed together, these elements show that topical drug development is a sophisticated integration of ADME science, formulation engineering, and clinical strategy. It is not enough to design a molecule that can cross the skin. Teams must design for the right target engagement profile in the skin, a safe systemic exposure, patient-friendly formulations, and robust preclinical research and clinical evidence that connect all these dots.
For scientists, clinicians, and business leaders looking to deepen their understanding, the webinar “Targeted Dermal Delivery: Integrating ADME Science in Topical Drug Development – Syngene International Ltd” is a useful reference point. It brings together practical case studies, quantitative data, and real-world experience on how to use ADME science, tirbanibulin experience, and dermal delivery strategy to bring topical therapies from concept to clinic.