Introduction to Synthetic DNA and RNA Interference
The COVID-19 pandemic is posing an unprecedented threat to public health, besides severely disrupting the global economy. Worldwide, millions have died, million more are infected and suffering loss of life and livelihood, and still there is no immediate solution in sight. Drug repurposing has been promptly attempted as a temporary measure, resulting in moderate success. While several biopharmaceutical companies are racing to develop drugs and vaccines, it may still take months or even years for a therapeutic remedy to emerge. Consequently, social distancing and quarantining infected individuals remain the recommended strategy to arrest the spread of the contagion. With the number of cases continuing to grow, extensive availability of diagnostic testing and rapid identification, becomes critical for the success of this approach.
Synthetic oligonucleotides have emerged as an essential modality for diagnostic and potential therapeutic purposes. The significant advantages of oligonucleotide-based strategies are that they can be quickly designed based on predictable Watson-Crick hybridization and synthesized with well-developed solid-phase synthesis chemistries. For example, immediately after the Chinese Center for Disease Control and Prevention identified severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) as the cause of the new disease, the genomic sequence was made publicly available. In the absence of specific antibodies for this novel virus, polymerase chain reaction (PCR) based diagnostic tests using oligos were promptly developed based on the genomic data.
This article focuses on the application of synthetic oligonucleotides in combating the COVID-19 pandemic. We will discuss the diagnostic tests to detect the infection and the role of oligonucleotides in these tests. We will also briefly touch upon potential strategies on how oligonucleotides can be used to treat infected patients, and arrest further progression of the virus.
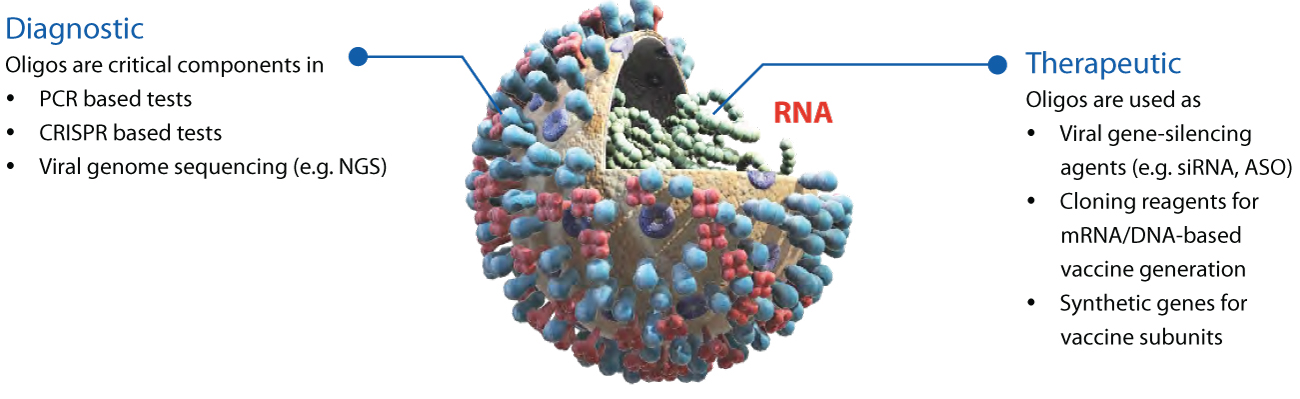
This article focuses on the application of synthetic oligonucleotides in combating the COVID-19 pandemic. We will discuss the diagnostic tests to detect the infection and the role of oligonucleotides in these tests. We will also briefly touch upon potential strategies on how oligonucleotides can be used to treat infected patients, and arrest further progression of the virus.
Oligonucleotides for COVID-19 Diagnostics and Therapeutics
Commercially available COVID-19 tests presently can be classified into two major categories1 . The first category, also called molecular tests, relies on PCR-based techniques to detect viral RNA. The second category depends on serological and immunological assays that detect antibodies generated in infected individuals.
Molecular Tests: RT‑PCR Testing and Solid‑Phase Oligonucleotide Synthesis
PCR-based Methods
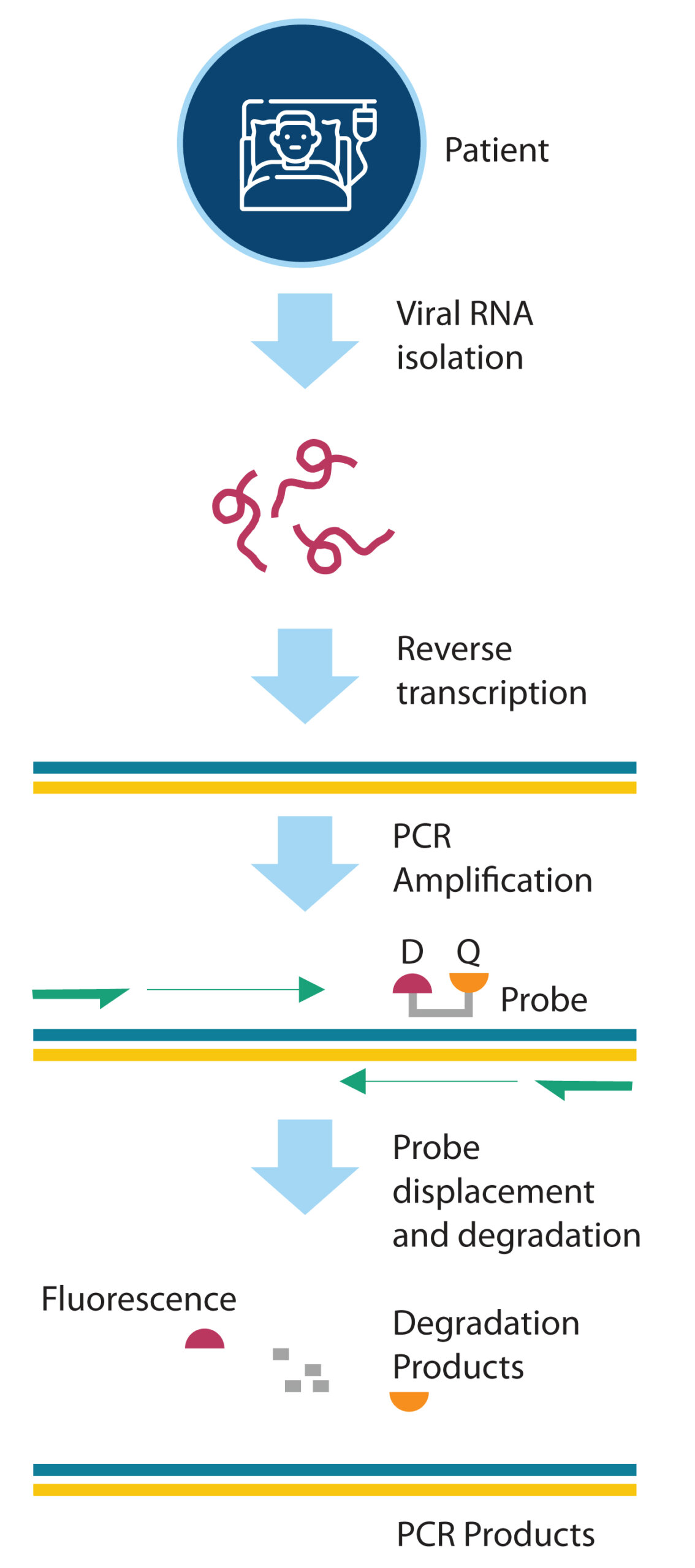
In the PCR-based methods, synthetic oligonucleotides are directly used to amplify and detect the viral RNA. There are several variations of the PCR-based techniques, and they are continually evolving to include automation, improve accuracy, and reduce assay duration. The majority of the molecular tests performed to date are based on real-time reverse transcription PCR (RT-PCR) technique that targets different regions within the SARSCoV-2 virus genome.2 In the RT-PCR method (Figure 2), DNA oligonucleotide primers recognize and bind to the viral genomic RNA, if present in the isolated RNA from the patient sample. The reverse transcriptase enzyme then converts the RNA to the short complementary DNA (cDNA). The DNA polymerase subsequently amplifies this cDNA template. The assay also contains reporter fluorescent dye (D) and quencher (Q)-labeled oligonucleotide probes that bind with the cDNA. The proximity of the quencher molecule keeps the fluorescent intensity at a low level.
During PCR amplification, the probe gets displaced and digested by the polymerase to provide measurable fluorescence in real-time. If the fluorescence signal crosses a certain threshold level, the test is deemed positive. Although the RT-PCR test is considered the ‘gold standard’ for COVID-19 detection, it is laborious, requires expensive instrumentation, and takes days to get the result. These practical limitations are seriously limiting mass screening, which is critical to combat the contagion. Consequently, other versions of the PCRbased tests have been developed that rely on isothermal amplification technologies. Unlike RT-PCR tests, where sophisticated thermal cyclers are required, isothermal tests are designed to work with simple incubation at a specific temperature.
Examples of such tests are, reverse transcription loop-mediated isothermal amplification (RT-LAMP), transcriptionmediated amplification (TMA), and rolling cycle amplification (RCA). Notably, all these methods employ synthetic oligonucleotides as essential reagents. For instance, the RT-LAMP technologybased COVID-19 test utilizes a set of DNA oligonucleotide primers and molecular beacons or an intercalating dye, along with reverse transcriptase and DNA polymerase enzymes to rapidly (less than an hour) diagnose the infection at the point-of-care itself.3 While isothermal methods greatly simplify COVID-19 tests, wide adaptation is yet to happen on a global scale.
CRISPR-based Methods CRISPR
(Clustered Regularly Interspaced Short Palindromic Repeats) geneediting technology has revolutionized biotechnology in every possible way, and the COVID-19 test is not an exception. Several versions of the CRISPR-based tests, such as SHERLOCK, DETECTR, CREST, and FELUDA, have been developed with some variations.4 In general, these methods use DNA-primers for reverse transcription and isothermal amplification of the viral RNA. In the next step, a synthetic RNA (e.g., Trans-activating CRISPR RNA or tacrine) recognizes and binds to the viral-derived sequence. The methods also contain Cas proteins (e.g., Cas12a for SHERLOCK) that cleave the viral-derived target sequence or reporter oligonucleotide molecules. The outcome of the test can be recorded with lateral-flow strips or fluorescence signals. CRISPR-based tests hold immense potential because they are inexpensive, rapid, and more specific than traditional tests. Other Methods In addition to the PCR and CRISPR-based methods, other molecular tests can recognize the presence of SARS-CoV-2 infection. Most of these methods also rely on synthetic oligonucleotides as one of the critical components. For example, nextgeneration sequencing (NGS) techniques have been developed for direct, highthroughput, and specific identification of infections in a variety of patient specimen types. Notably, the first sequencing and identification of the SARS-CoV-2 virus relied on NGS technology and used synthetic oligonucleotides as sequencing adapters.5 Since direct sequencing can often be challenging because of the low abundance of viral sequences, ampliconbased methods have been reported. Such technology first uses PCR-reactions and synthetic DNA primer sets to generate amplicons, which are further sequenced to detect the virus infection.
Molecular Tests: RNA Interference and Oligonucleotide Therapeutics
While significant effort is underway, an effective antiviral strategy to treat SARS-CoV-2 infection does not exist to date. Along with the small (e.g., antiviral drug) and large molecule (e.g., vaccines, antibodies) – based approaches, oligonucleotide therapeutics signify an emerging strategy to combat the contagion.7 Synthetic oligonucleotides can inhibit gene expression by several avatars such as antisense oligos (ASOs) and silencing RNAs (siRNAs). They directly bind with the complementary disease-causing target gene sequence and inhibit the corresponding protein synthesis, either by blocking the ribosome access or by enzyme-assisted destroying of the target mRNA. Such oligonucleotides have been used for antiviral applications with some promising success.8 For example, the United States Food and Drug Administration approved the ASO fomivirsen (Brand name Vitravene) in 1998 to treat cytomegalovirus (CMV) retinitis — an infection of the retina in HIV patients that can lead to blindness. Similarly, ASOs and siRNAs have been proposed, to target the critical component of the virus itself, e.g., the spike protein. In the past, morpholino ASOs have been shown to inhibit some other corona virus classes.9 Several companies are currently working on applying such therapeutic oligonucleotides for many viral diseases, including COVID-19. Unlike small molecules, oligonucleotide drug design often follows established rules. Therefore, the success of these approaches will lead to a sustainable therapeutic strategy for the current and future outbreaks. As discussed before, synthetic oligonucleotides are necessary reagents in recombinant molecular biology techniques to generate proteins and mRNAs. Along with direct applications as siRNAs or ASOs, oligonucleotides thus have found their applications as primers in potential DNA/mRNA-based vaccine development. Advances in gene synthesis using oligonucleotide technologies can rapidly generate viral antigen subunit vaccines, avoiding the use of dangerous viral pathogens. Nonetheless, considering the myriad ways the scientific community is trying to tackle the COVID-19 pandemic, applications of oligonucleotides are likely to spread in newer directions.10
What Can Syngene Offer in Oligonucleotide Therapeutics and RT‑PCR Testing?
Syngene’s oligonucleotide facility was established in 2014 as a process development lab capable of producing 100 mg to 6 g of therapeutic oligonucleotides in a non-GMP environment. The GMP manufacturing facility which can synthesize 6 g to 135 g of oligonucleotides, started functioning in 2017. Over the years, Syngene has established advanced skills to handle a wide range of therapeutic oligos, including all kinds of sugar, backbone, and base – modifications, all common end -modifications, as well as natural DNA and RNA oligonucleotides. Currently, Syngene houses one of the best oligonucleotides centers in Asia, and the only one in India, to support GMP-grade oligonucleotide synthesis on the indicated scale. The competitive advantage of Syngene’s service is an independent-yet-integrated quality management system that has been approved by several national and international regulatory agencies
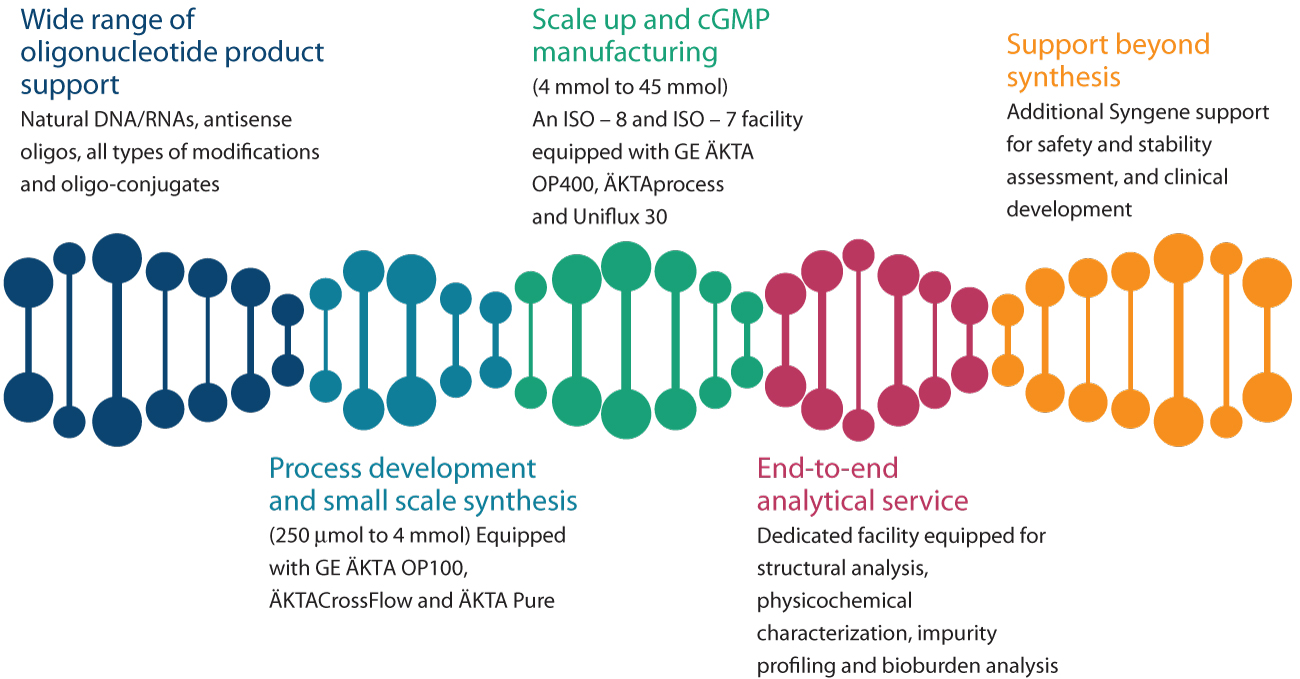
Along with therapeutic oligonucleotides, during the COVID-19 pandemic, the oligonucleotide facility was promptly repurposed to synthesize diagnostic oligonucleotides as well. The facility since then has been supplying primer and dual-labeled probes for RT-PCR testing kits. Syngene offers guaranteed high-purity oligonucleotides with competitive turn-around times, and will be delighted to extend its service to potential clients. For more information about our oligonucleotides service, write to bdc@syngeneintl.com.
About the author








