Introduction to Biopharma R&D Innovation Challenges
The pharmaceutical industry is facing a sustainability crisis due to dwindling ROI on research and development. Even though the top 20 pharmaceutical companies invested 20.9% of topline revenues into R&D last year, the internal rate of return (IRR) for pharma R&D is already below the cost of capital and is projected to hit zero.
Thus, there is a pressing innovation challenge in the industry today. Organizations are seeking novel strategies to manage expenditure, reduce development timelines and increase returns. Many options have been considered in the quest to bring innovation back, such as academic collaborations, open-source ideation, and M&A. However, two strategies stand out in terms of efficacy and ease of adoption: a) Establishing external R&D partnerships and b) Virtualization and outsourcing.
Syngene also offers a dedicated center model of outsourcing that provides benefits of improved operational efficiency, opportunity to become part of an innovation network, multi-tiered governance system, access to world-class infrastructure and facilities, and more.
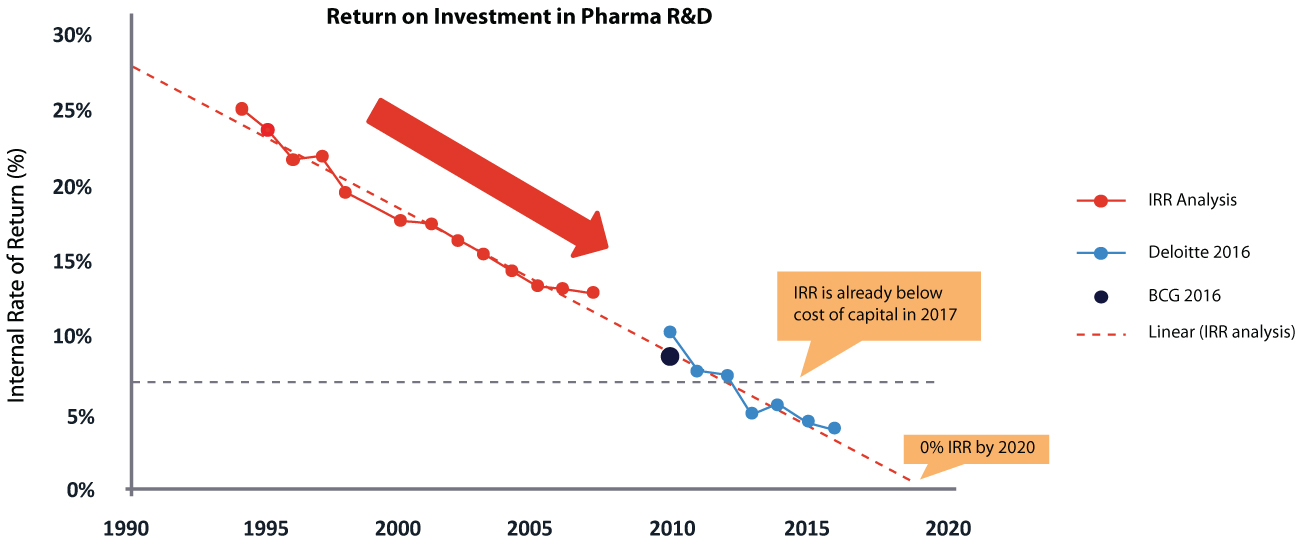
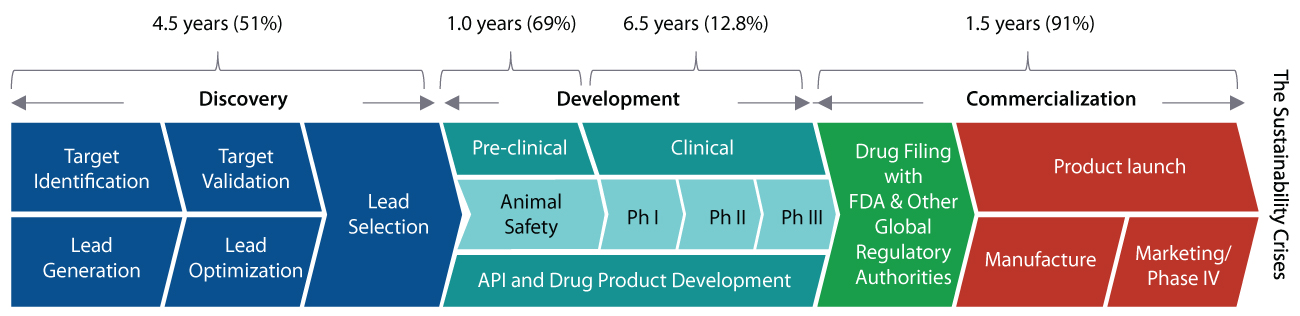
According to studies, it takes ~14 years and over US$2.6 Bn to bring a product to market at an overall success rate of just ~4.1%. Recent data also shows an increase in timelines for Pre-clinical (+17 %, 2004–2012) and Phase I development (+58 %, 2004–2012), leading to higher costs.
Thus, there is a pressing innovation challenge in the industry today. Organizations are seeking novel strategies to manage expenditure, reduce development timelines and increase returns. Many options have been considered in the quest to bring innovation back, such as academic collaborations, open-source ideation, and M&A. However, two strategies stand out in terms of efficacy and ease of adoption: a) Establishing external R&D partnerships and b) Virtualization and outsourcing.
How Outsourcing Drives Biopharma Innovation
As pharmaceutical companies seek ways to improve research ROI, outsourcing has shifted from being purely a cost arbitrage tactic to an innovation strategy. When organizations mature through the outsourcing lifecycle, the number of outsourced services and the duration of engagements increase. This is typically accompanied by evolution from component play -> cluster play -> integrated play -> dedicated play. Along with this evolution comes a shift in focus from short term gains to building a knowledge continuum with the outsourcing partner. The integrated play and dedicated play emerge as preferred partnership models when organizations start realizing innovation dividends from close integration between outsourced teams and their internal research.
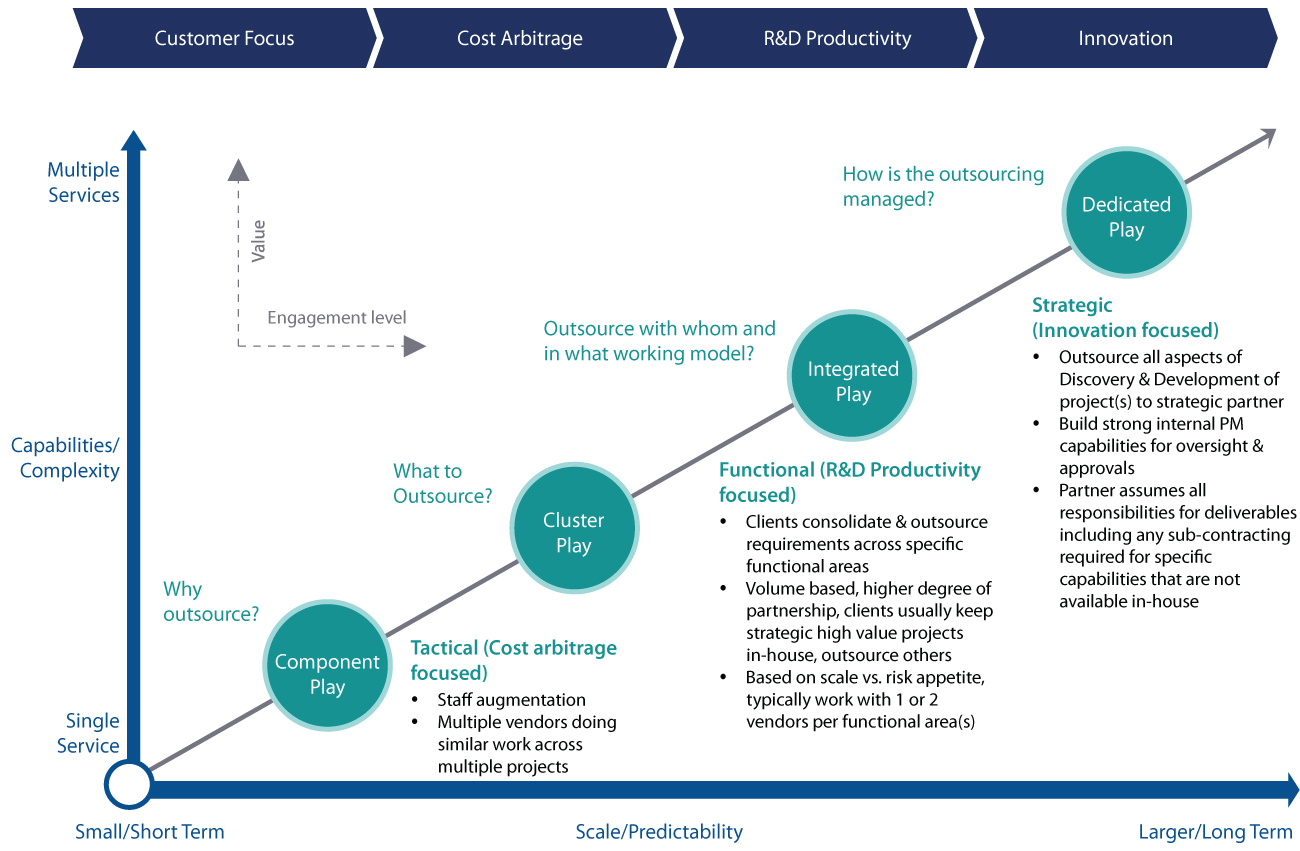
An innovation-focused integrated/dedicated play involves outsourcing all aspects of a research program to a strategic partner. The outsourcing partner assumes responsibility for all deliverables including any sub-contracting for capabilities that are unavailable inhouse. A partner with end-to-end capabilities from discovery to commercial manufacturing is essential for such a model to be effective.
Full-Scale Biopharma R&D Integration Model
Syngene has had significant success in deploying integrated and dedicated center models for its clients. One such engagement was with a Japanese client on an integrated program spanning over 17 years. This collaboration on a specific molecule began when the client approached Syngene for production at a sub 1Kg scale. Over the years, the scale of the engagement went from <1Kg to >1000Kg, as we followed the molecule through tox, FIH, phase 2, phase 3 and eventually commercial manufacturing.
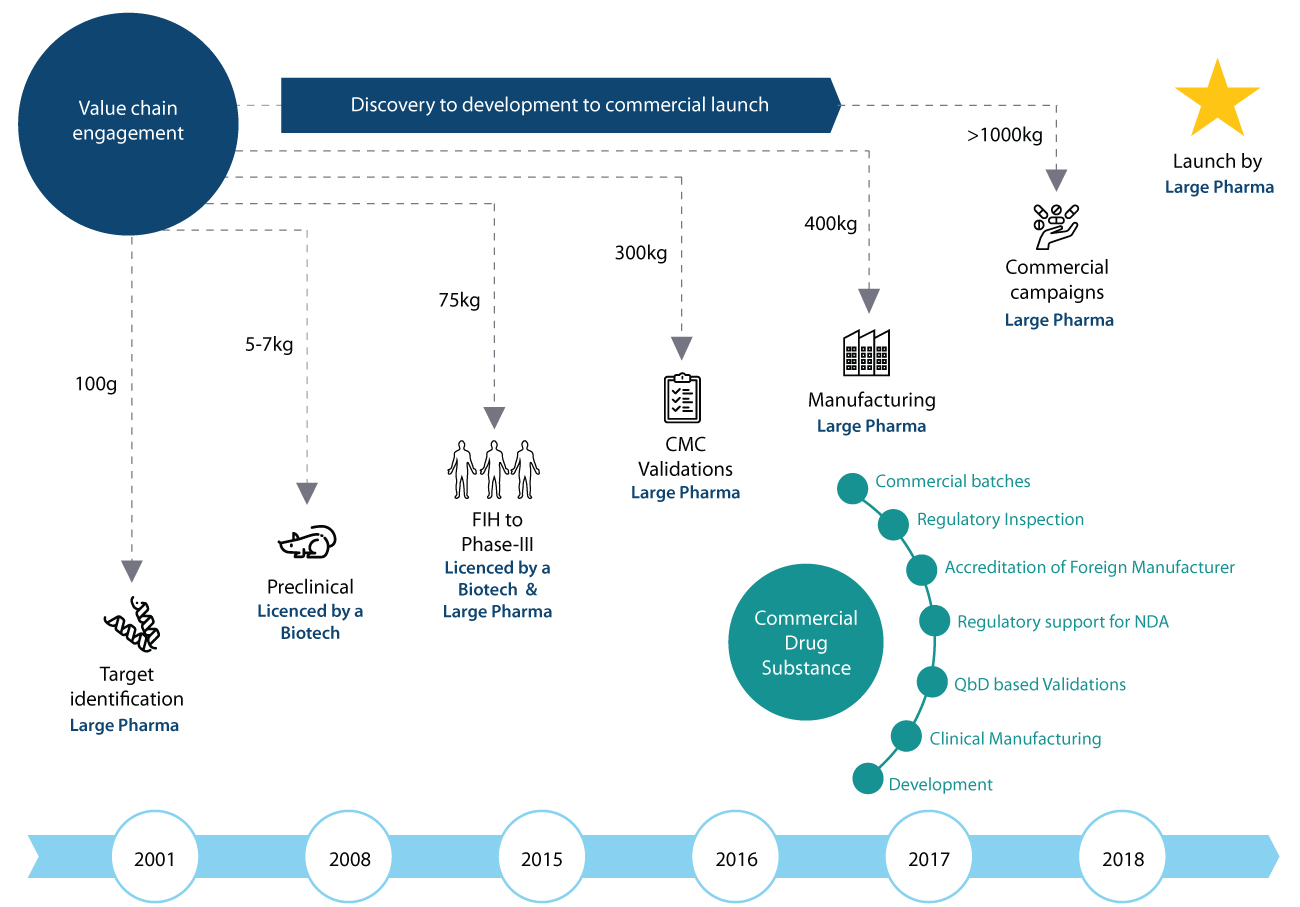
This collaboration was particularly noteworthy due to the contribution made in terms of innovation and process optimization. Even before reaching commercial production, Syngene was able to reduce the no. of steps needed for the synthesis from 17 to 12. Similarly, on one of the steps, the yield was enhanced from 5% to 23%. We were even able to build second sourcing vendors at a much earlier stage in the drug lifecycle, resulting in reduced costs and improved efficiency.
Dedicated Centers for Integrated Outsourcing Solutions
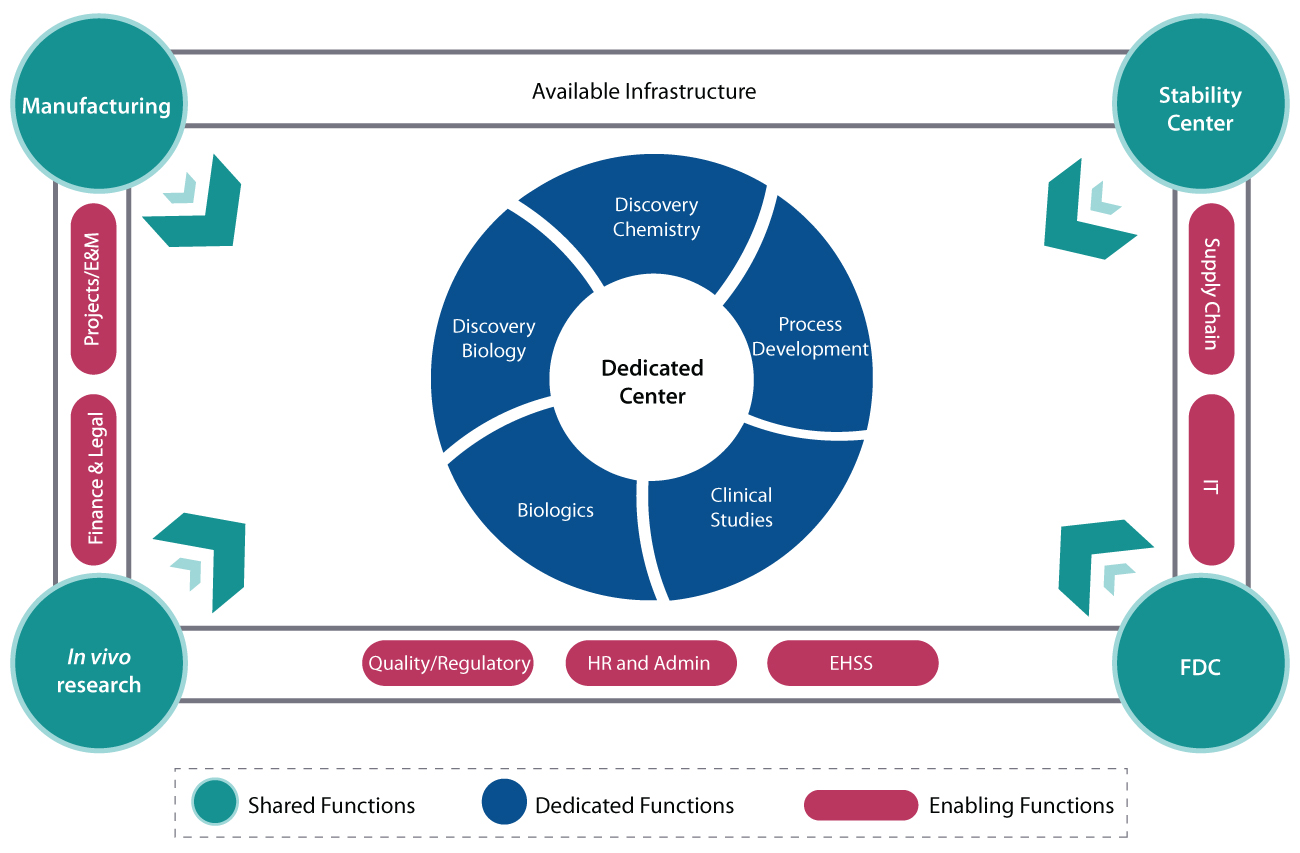
Under the dedicated center model at Syngene, depending on the program, the sponsor organization will get a set of ring-fenced personnel dedicated to the program as well as access to enabling functions such as HR, facilities management, quality regulation, etc. The innovator company can work directly with the staff at Syngene, while also getting logistics support and audit support from the strong in-house program management team. The dedicated model is the equivalent of having access to a full-fledged pharmaceutical company entirely housed in a single campus, with capabilities across small and large molecules. What this means is that even niche capabilities that are not part of the dedicated center setup will be available on an as-needed basis as part of Syngene’s shared analytical facility
BMS Dedicated Center at Syngene: Pharma Partnership Success
Bristol-Myers Squibb Dedicated Center at Syngene (BBRC)
Established in 2007, the BBRC is a dedicated research center at Syngene for Bristol-Myers Squibb (BMS). It operates as a cost-effective, fully integrated research environment focused on delivering high-quality scientific outcomes.
Research Focus Areas:
Immunoscience
Cardiovascular diseases
Arthritis
Endometriosis
Neuropsychiatric pain and depression
Key Success Factors:
IP Protection
All intellectual property generated is assigned to BMS.
This ensures seamless information exchange across partnering companies.
Scalability
The center has expanded over time, adding capital expenditure (CAPEX).
CAPEX investments have been converted to operational expenditure (OPEX), enabling consistent pipeline delivery.
High-Quality Talent Pool
Draws from the combined talent of BMS, Biocon, and Syngene.
Supported by robust training programs and well-structured employee policies.
Impact and Achievements:
Low Attrition Levels
Reflects strong employee engagement and satisfaction.
Largest BMS R&D Center in Asia
A testament to its scale and strategic importance.
Drug Development Milestones
Delivered 10 drug candidates for further development.
Generated new compounds for first-in-human studies.
Contract Extension
The partnership has been extended until 2026.
Reducing Drug Development Costs with Syngene's Dedicated Centers
The dedicated model delivers manifold benefits for the service seeker:
• 20-25% improvement in operational efficiency per FTE (synthesis of molecules, TAT, assays completed, etc.)
• Employees identify with values of the client organization and become part of an innovation network
• Multi-tiered governance system, direct access to scientists and a framework for leadership level decision making
• Infrastructure and facilities including data security, canteen, supplies, and wellness Amidst challenges in terms of pipeline, ROI, and innovation, the dedicated center model of outsourcing allows pharmaceutical companies to place strategic bets to achieve long term goals of their research programs.
An outsourcing partner such as Syngene, with over 25 years of expertise in advancing the innovation agendas for clients across the globe, can help achieve these goals in a time-bound and cost-effective manner.
About the author








