Gene editing has moved rapidly from a breakthrough laboratory tool to a central pillar of modern therapeutic innovation. Over the past decade, advances in programmable nucleases and RNA-guided systems have transformed how scientists manipulate DNA with high precision. Currently, gene editing is no longer defined only by CRISPR technologies. A new generation of editing platforms including base editing and prime editing is expanding the possibilities of genome engineering, while also addressing long standing concerns such as crispr off-target effects and editing efficiency.
For drug developers, these technological advances are reshaping the landscape of therapeutic discovery and development. Gene editing now supports multiple stages of the biopharma pipeline, including target validation, disease modeling, cell engineering, and development of gene and cell therapies. At the same time, the growing complexity of editing technologies has increased the importance of specialized research partners capable of integrating biology, data science, and advanced screening platforms.
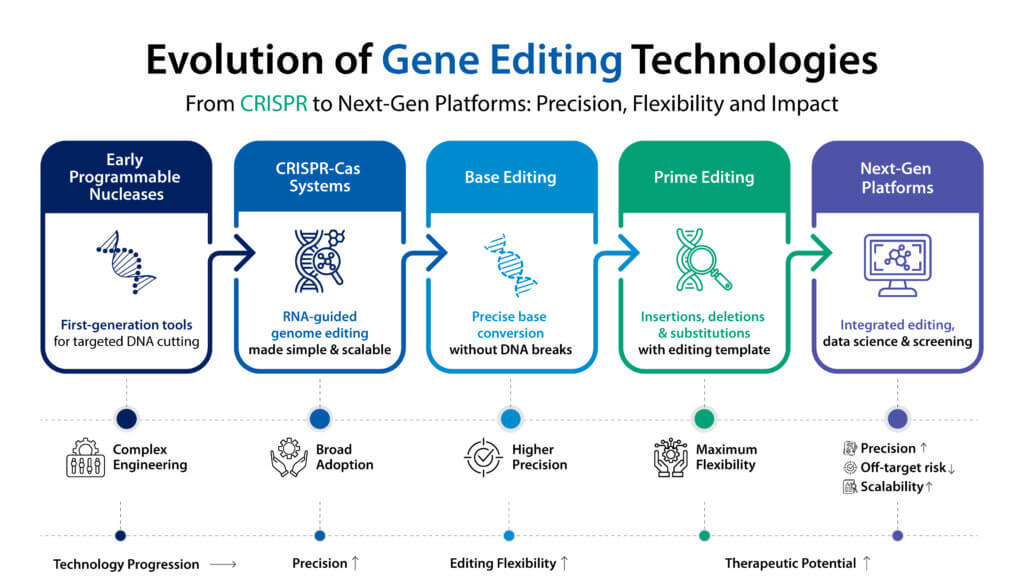
The evolution of gene editing technologies
The modern era of gene editing began with programmable nucleases such as zinc finger nucleases and TALENs. However, the introduction of CRISPR-Cas systems dramatically simplified genome engineering by enabling sequence specific targeting through guide RNA molecules. CRISPR-based gene editing quickly became widely adopted across academia and industry due to its speed, scalability, and adaptability.
Despite its transformative impact, early CRISPR-based editing approaches relied on creating double strand DNA breaks, which are then repaired by cellular mechanisms. While effective, this process can introduce unintended insertions or deletions and may contribute to CRISPR off-target effects in certain genomic contexts.
To address these challenges, researchers have developed editing platforms that modify DNA without introducing double strand breaks. These next generation technologies are expanding the precision and versatility of gene editing and are now being explored across multiple therapeutic modalities.
Base editing and the refinement of precision
Base editing represents one of the most important innovations in the evolution of gene editing. Instead of cutting both strands of DNA, base editing chemically converts one nucleotide into another at a specific genomic location. This enables precise correction of point mutations that are responsible for many genetic diseases.
In practical terms, base editing systems combine a modified CRISPR enzyme with a nucleotide converting enzyme. The CRISPR component directs the complex to a specific DNA sequence, while the catalytic domain performs the targeted base conversion. Because no double strand break is created, base editing can significantly reduce unwanted insertions and improve editing outcomes in certain applications.
Several therapeutic programs are now exploring base editing strategies for conditions driven by single nucleotide mutations, including hematologic disorders and inherited metabolic diseases. For pharmaceutical companies, these capabilities expand the toolbox of gene editing approaches available during both early research and translational development.
Prime editing technology and its expanding role
Among the newest innovations in the field, prime editing technology offers a highly versatile approach to genome modification. Prime editing builds on the CRISPR framework but uses a modified Cas enzyme paired with a specialized guide RNA that contains the desired genetic change.
This system enables a wider range of DNA modifications, including insertions, deletions, and base substitutions, without requiring double strand breaks. Because of this flexibility, prime editing technology is often described as a “search and replace” method for DNA sequences.
From a drug discovery perspective, prime editing has significant potential. It allows researchers to model complex genetic variants with high precision, which is particularly valuable in understanding disease mechanisms. Prime editing also opens the door to correcting mutations that were previously difficult to target using earlier gene editing platforms.
However, prime editing systems are still being optimized for efficiency, delivery, and scalability. The development of improved editing constructs, optimized guide RNA design, and advanced screening workflows continues to be an active area of research.
Addressing CRISPR off-target effects in therapeutic development
As gene editing moves toward clinical applications, understanding and mitigating CRISPR off-target effects remains a central focus. Off-target activity refers to unintended DNA modifications occurring at genomic sites that resemble the intended target sequence.
While advances in guide RNA design and improved Cas variants have reduced off-target risks, comprehensive characterization of editing outcomes remains essential during therapeutic development. Genome wide sequencing approaches, computational prediction models, and functional assays are now commonly used to evaluate editing specificity.
In addition, many development programs now combine gene editing with advanced computational analysis. Techniques such as large scale CRISPR screening and systems biology modeling are helping researchers better understand gene networks and identify optimal editing targets. These integrated approaches contribute to more reliable target discovery and validation strategies across therapeutic programs.
Insights from target identification and validation workflows are increasingly used to determine whether gene editing represents the most appropriate strategy for modulating a disease pathway. Such approaches combine genomic data, functional screening, and predictive analytics to support decision making early in the discovery pipeline.
Expanding applications across the biopharma pipeline
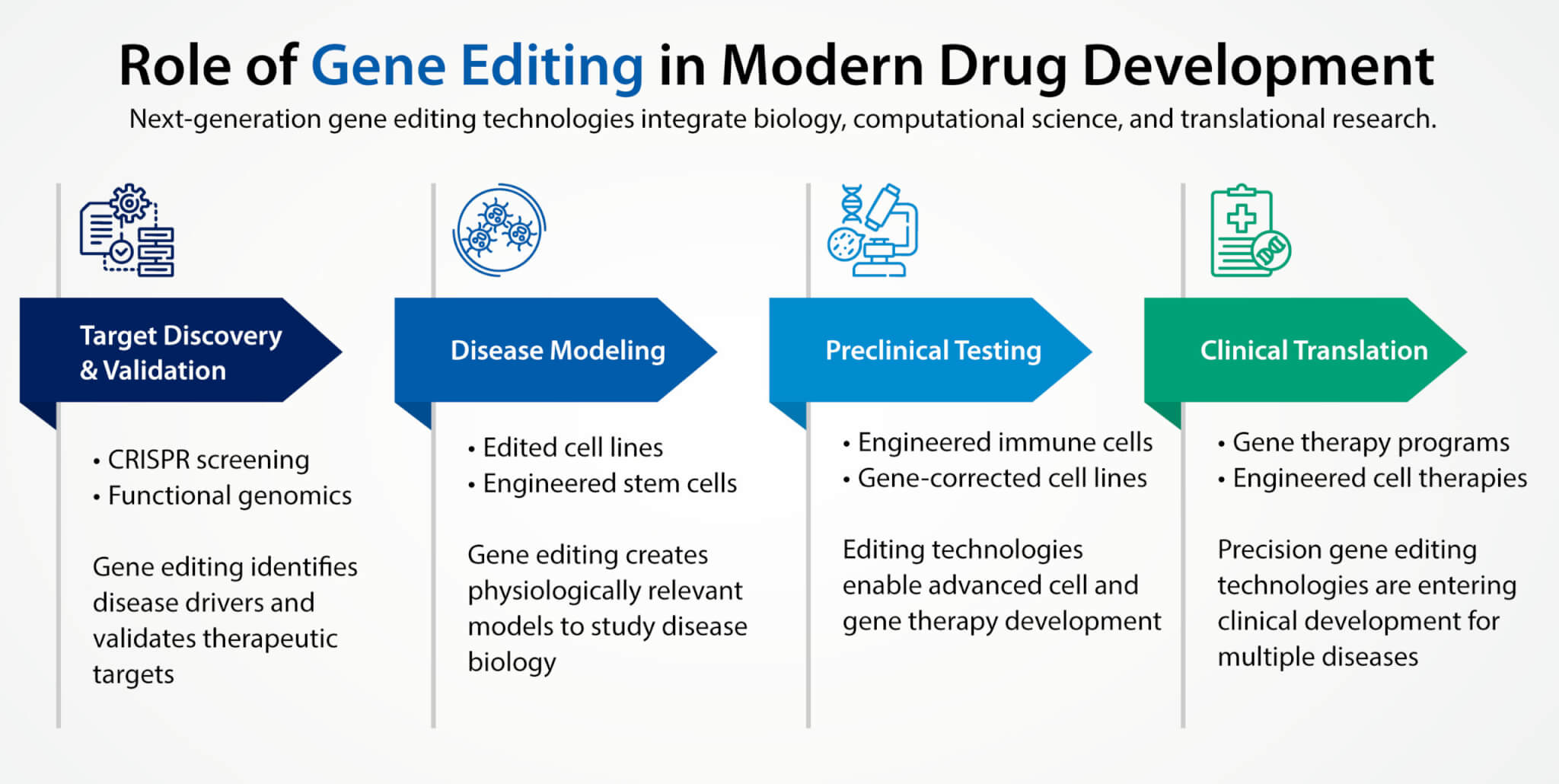
The role of gene editing extends far beyond the development of gene therapies alone. In modern drug discovery programs, gene editing technologies are widely used to generate cellular models, engineer immune cells, and validate therapeutic targets.
Figure 2. Gene editing technologies support key stages of drug development, including target discovery, disease modeling, therapeutic engineering, and clinical translation.
For example, gene editing can create disease specific cell lines that reproduce key genetic mutations observed in patients. These models allow researchers to study disease biology more accurately and test candidate therapeutics under controlled conditions.
Similarly, gene editing technologies are playing a central role in the development of cell therapies. In engineered immune cell platforms, editing strategies are used to improve persistence, reduce immune rejection, and enhance therapeutic activity. Advances in gene editing are also supporting the generation of engineered stem cell lines, which serve as powerful tools for disease modeling and drug screening.
In complex discovery programs, advanced cellular systems such as the iPSC platform capabilities enable researchers to combine stem cell biology with precise genome engineering. This integration supports the development of physiologically relevant models that better represent human disease mechanisms.
The CRO perspective on next generation gene editing
As gene editing technologies continue to evolve, the operational challenges associated with these platforms are also increasing. Effective implementation requires expertise across multiple disciplines including molecular biology, genome engineering, bioinformatics, and translational research.
Contract research organizations (CROs) increasingly play a critical role in supporting these complex workflows. From assay development and CRISPR screening to next generation sequencing analysis, CRO environments bring together the specialized infrastructure required for high throughput genome engineering.
Importantly, next generation gene editing platforms such as prime editing and base editing require careful optimization of experimental design, delivery systems, and validation strategies. The ability to integrate automated laboratory platforms with advanced analytics is becoming essential for achieving reproducible editing outcomes at scale.
In addition, regulatory expectations for gene edited therapies are evolving. Developers must demonstrate robust characterization of editing outcomes, including detailed analysis of potential off-target events. CRO partners with expertise in genomic analysis and assay development are therefore becoming increasingly valuable across the development continuum.
The future of gene editing innovation
By 2026, gene editing has clearly moved beyond a single technology platform. CRISPR-based approaches remain foundational, but the emergence of base editing and prime editing technology is expanding the capabilities of genome engineering in unprecedented ways.
These technologies are enabling more precise, flexible, and scalable editing strategies across a wide range of therapeutic areas. At the same time, advances in computational biology, sequencing technologies, and automated laboratory systems are accelerating the pace at which gene editing discoveries can move from research to clinical development.
For the biopharma industry, the next phase of gene editing innovation will likely involve integration across multiple technologies rather than reliance on a single platform. As researchers continue to refine editing accuracy and delivery systems, gene editing will play an increasingly central role in the development of next generation therapeutics.