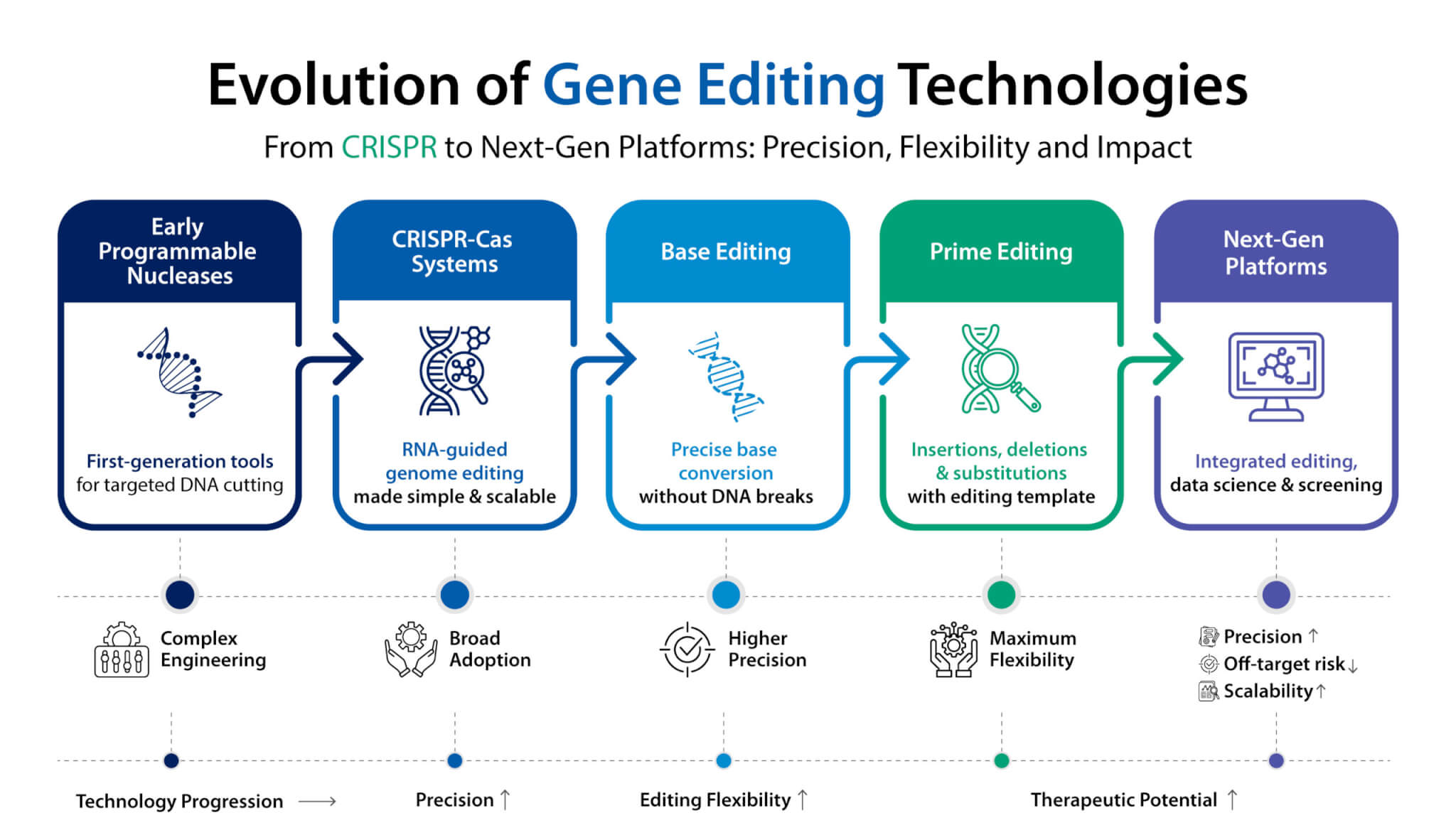
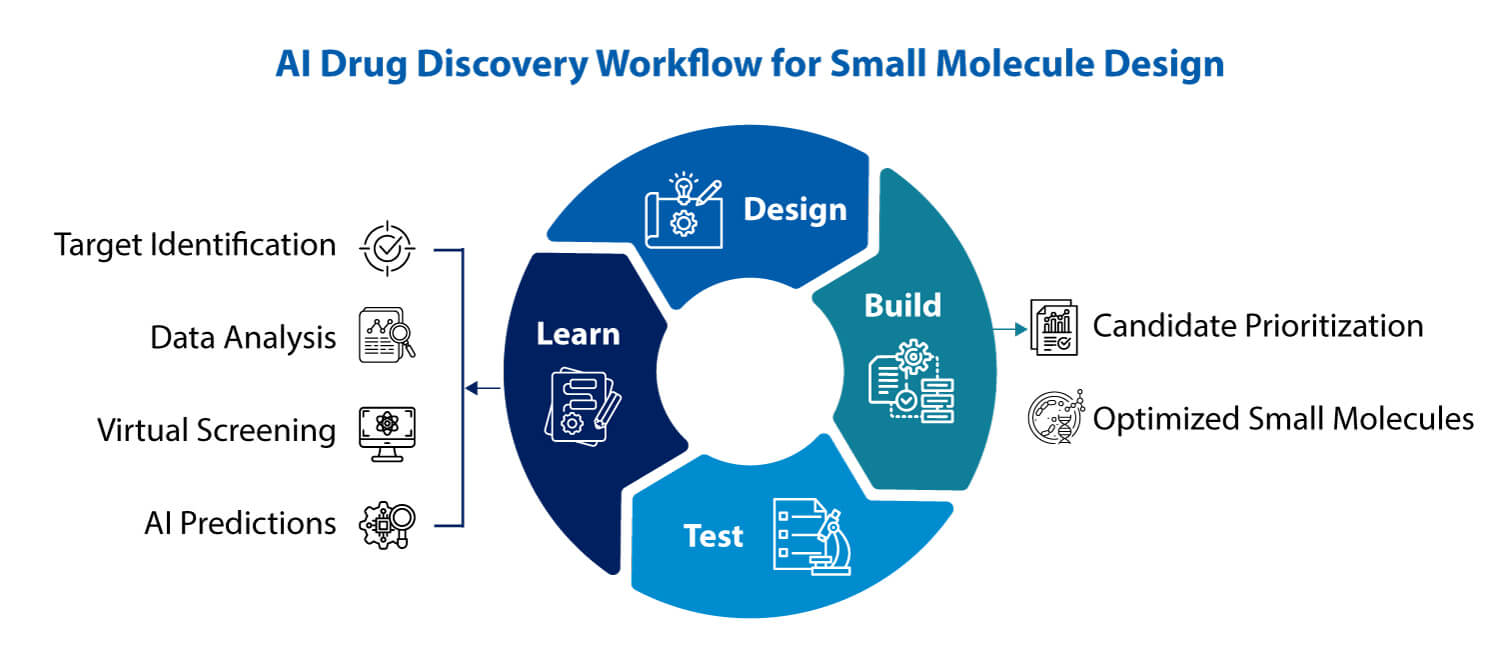
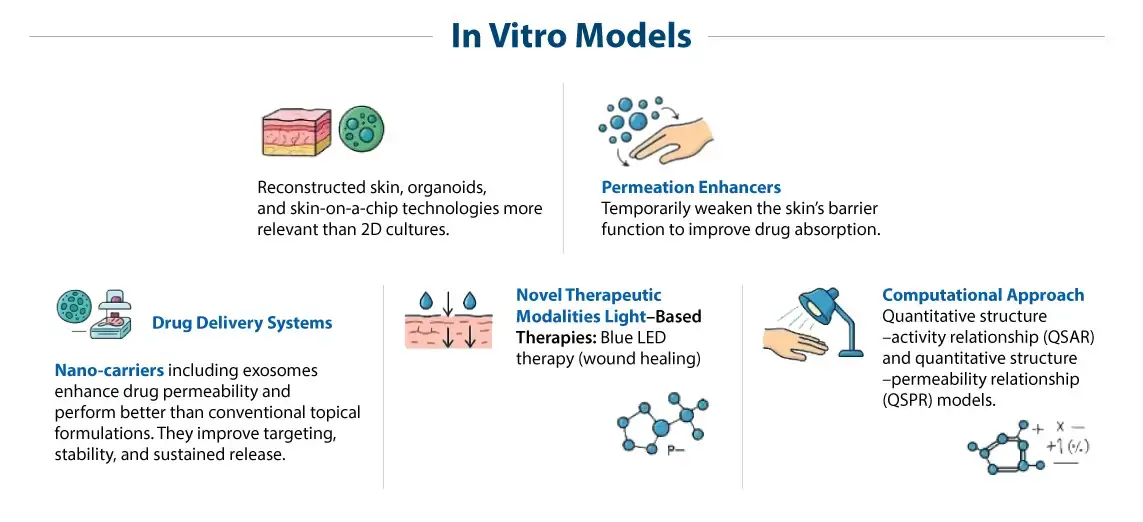
At Syngene, technology transfer is a seamless, cross-site collaboration, accelerating your journey from concept to commercialization. This video showcases the synergy between our Bangalore (BSEZ) and Mangalore (MSEZ) campuses, driven by robust systems and deep expertise. Our facilities are built for uncompromising quality, proudly GMP certified, 21 CFR compliant, and USFDA approved, ensuring global standards are met at every step. From advanced process and analytical research to meticulous engineering and safety protocols, we emphasize alignment, transparency, and cost-effective execution. Discover how Syngene’s commitment to scalable, economical, and safe processes, supported by our dedicated teams, translates into reliable outcomes. Partner with us for long-term manufacturing and supply solutions, backed by scientific alignment and operational synergy that truly puts science to work for your success.
- About Us
- Solutions
- Small Molecules
- Biologics
-
- Other Drug Modalities
- SynVent Integrated Drug Discovery
-
SynVent Integrated Drug Discovery
SynVent is Syngene’s platform for fully integrated therapeutic discovery and development across large and small molecules.

-
- Industries
- Emerging Biopharma
-
Emerging Biopharma
Emerging biopharma work at the forefront of science, often venturing into disease areas where little or no real-world data exists to work with or regulatory frameworks to work within.

-
- Dedicated Centers
-
Dedicated Centers
Our Dedicated Centers offer dedicated multi-disciplinary scientific teams, support personnel, and a tailormade ring-fenced and fire-walled infrastructure as per client specifications to support their R&D goals

-
- Center for Advanced Protein Studies (CAPS)
-
Center for Advanced Protein Studies (CAPS)
Centre for Advanced Protein Studies [CAPS] is a state-of-the-art advanced national facility located in the Syngene campus, Bangalore.

-
- Small Molecules
- Investors
-
- Careers
- Newsroom
- Insights & Resources
- Group Companies