End-to-end Oligonucleotide services: Early Discovery to cGMP Manufacturing
Antisense oligonucleotides (ASOs) show great promise in treating many devastating diseases and conditions due to their ability to target mRNAs which play a crucial role in protein synthesis. However, being complex molecules, they require sophisticated chemical synthesis processes, which can be time-consuming and expensive. Added to this are challenges of purification, stability, delivery, and adherence to strict regulatory standards during manufacturing to demonstrate the safety and efficacy of the products.
Syngene has significant expertise in oligonucleotides. We offer services ranging from discovery to cGMP manufacturing, enabling rapid development at lower costs. We specialize in chemically synthesized high-quality modified and non-modified custom oligonucleotide (DNA/RNA), siRNA (single & double-stranded, miRNA (modified and non-modified), and antisense oligonucleotides, at various scales with rapid turnaround time. Together with our state-of-the-art oligonucleotide facility, a highly qualified team of scientists, and global client experience, we have all it takes to become the partner of choice for companies looking to accelerate their oligonucleotide programs.
Our service offerings
| Natural DNA oligonucleotides | Phosphothioates and morpholinos |
| Natural RNA oligonucleotides | All types of oligo conjugates |
| Aptamers and siRNAs | Fluorescent oligos and molecular beacons |
| All types of end modifications | Custom modification development |
| All types of base modifications | Enzymatic oligomerizations |
| All types of sugar modifications |
Figure 1: Types of oligonucleotides and services supported
Flexible synthesis scale: Discovery to cGMP manufacturing
Discovery-scale synthesis
We offer synthesis support ranging from 50 nmole to 200 µmole. For discovery-scale synthesis, we use a K&A H16 oligo synthesizer with a cycle time of 5.5-6.0 min for 16-column synthesis.
Small-scale synthesis
We offer synthesis support ranging from 250 mmol to 4 mmol. Our small-scale synthesis is equipped with GE ÄKTA OP100, ÄKTACrossFlow, and ÄKTA Pure facilities.
cGMP manufacturing
We offer synthesis support ranging from 4 mmol to 45 mmol. Our cGMP manufacturing facility is an ISO-8 and ISO-7 facility equipped with GE ÄKTA OP400, ÄKTA process, and Uniflux 30.
Dedicated Analytical facility
Our state-of-the-art, end-to-end Analytical facility specializes in structural analysis, physicochemical characterization, impurity profiling, and bioburden analysis. With these facilities and equipment, we offer comprehensive support for oligonucleotide synthesis, analysis, and manufacturing, ensuring high-quality, efficient, and cost-effective services.
Syngene's Center of Excellence for oligonucleotides
The Syngene oligonucleotide team has a wealth of experience synthesizing and profiling ASOs. Our Center of Excellence for oligonucleotides offers end-to-end services ranging from Discovery to Development and cGMP Manufacturing. The figure below shows the time efficiencies that biopharma companies can gain if partnering with CROs/CDMOs like Syngene having an integrated workflow.
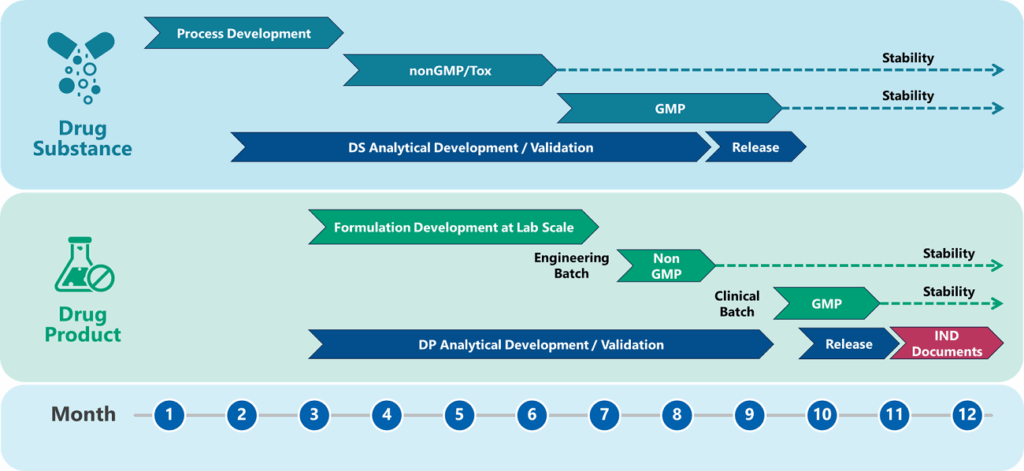
Accelerating Oligonucleotide supply by Integrating Drug Substance (DS) and Drug Product (DP)
Syngene has used its wealth of experience and knowledge in Oligonucleotide API development and manufacture (DS), as well as Clinical / Commercial drug product (DP) development and manufacturing, to create a fully integrated program for clinical / commercial supply. The figure 5 shows how we can perform development workflows in parallel in the same facility, and under the same quality system. The enhanced knowledge sharing and consolidation of activities speeds up the development cycle to provide clinic or commercial ready Oligonucleotide Drug Product.
The Syngene advantage
- Proven track record in oligonucleotide synthesis, including GMP synthesis and biological characterization
- Center of Excellence for end-to-end Discovery, Development, and Manufacturing services
- Comprehensive and fully integrated services for rapid development at lower costs
- Flexibility in synthesis scale with fast turnaround times
- Dedicated Analytical facility for structural analysis, physicochemical characterization, impurity profiling, and bioburden analysis






