A sensitive MSD-based ADA bioanalytical assay enabled reliable immunogenicity assessment for a clinical RNAi therapeutic, supporting ongoing clinical development while protecting a highly sensitive biotinylated drug reagent
At a glance
A global biotech developing a small interfering RNA (siRNA) therapeutic engaged Syngene and its advanced bioanalytical laboratory solutions for biologics to develop and run an anti-drug antibody (ADA) assay for clinical studies as part of its large molecule bioanalysis strategy. Using the Meso Scale Discovery (MSD) platform, Syngene established an advanced bridging immunogenicity method with sensitivity below 5 ng/mL and drug tolerance that allowed 10 ng/mL ADA to be measured in the presence of 10 µg/mL drug. Method optimization focused on preserving a sensitive biotinylated drug reagent and on controlling variability. The final assay delivered precise, reproducible ADA data suitable for regulated clinical development and future regulatory submissions.
Background and sponsor context
RNA interference is now an important modality for targeting disease-related genes. The sponsor was advancing a systemic siRNA therapeutic and needed a clear view of potential immunogenicity, including ADA incidence and magnitude, to meet regulatory expectations for its global clinical development program.
An MSD-based ADA assay was required that could detect low-level responses, remain tolerant to circulating drug, and be robust enough to support multiple clinical studies across the clinical development lifecycle.
Scientific and operational challenge
The immunogenicity bioanalytical strategy relied on a biotinylated form of the siRNA drug as a capture reagent, with labeled protein used for ADA detection. While this design was specific and precise, the biotinylated drug was highly sensitive to handling and conjugation.
Preparing frequent new conjugate lots risked loss of activity, inconsistency, and high consumption of a specialized reagent. At the same time, the assay had to maintain sensitivity at low ADA concentrations and cope with samples containing significant levels of therapeutic. Dilution patterns also had to be chosen carefully so that small pipetting differences did not add variability during routine clinical bioanalysis runs.
Syngene’s approach
Assay design and platform use
Syngene built a bridging ADA assay on the MSD electrochemiluminescence immunoassay platform. ADA in the sample bridges the biotinylated drug and a ruthenium-labeled Protein A/G detection reagent. The complex is captured on streptavidin-coated plates and read on the MSD instrument, giving a specific signal for anti-drug antibodies within a ligand-binding assay framework.
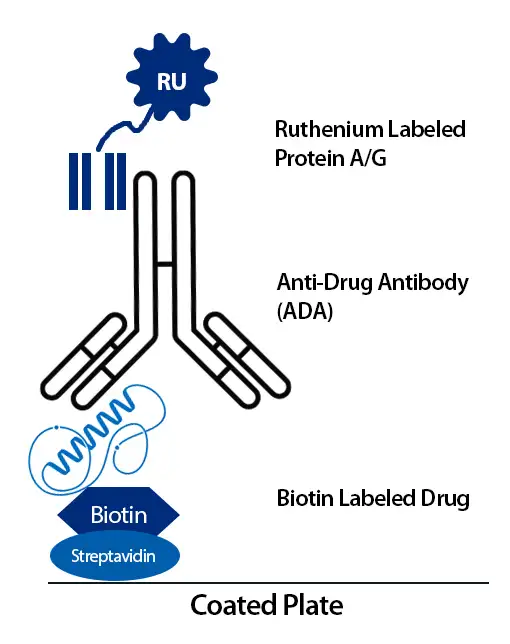
Development focused on achieving an appropriate signal-to-noise profile, a wide working range, and minimal hook effect at higher ADA levels.
Protecting the biotinylated drug reagent
To manage the sensitive conjugate, Syngene minimized repeated conjugation steps and defined conditions that preserved the integrity of the biotinylated drug. Stable, ready-to-use lots were prepared so that clinical runs relied on consistent reagents rather than frequent fresh conjugations, reducing variability and risk in regulated bioanalysis.
Optimizing sensitivity, linearity, and drug tolerance
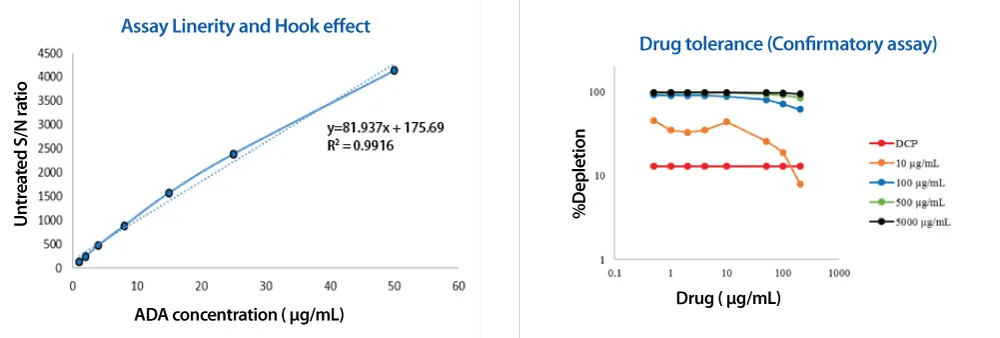
The optimized assay reached an MSD platform sensitivity of less than 5 ng/mL ADA. Linearity studies confirmed proportional responses across the intended ADA range, and hook-effect assessments showed reliable performance at higher concentrations.
Drug tolerance was tuned so that 10 ng/mL ADA could be measured in the presence of 10 µg/mL drug. This balance allowed detection of treatment-emergent ADA even when residual therapy was still present in circulation, supporting confident immunogenicity risk assessment.
Setting dilution patterns to reduce variability
Sample and reagent dilutions were adjusted to use higher microliter volumes. Working at slightly larger volumes reduced the impact of pipetting error and day-to-day variation, which was important when processing large numbers of clinical samples under regulated GxP conditions.
Outcome and impact
The final ADA assay met the performance needs for clinical development of the siRNA therapeutic. It delivered:
Sensitive detection of ADA responses (LOD < 5 ng/mL).
Demonstrated drug tolerance at 10 ng/mL ADA in 10 µg/mL drug.
Stable behavior across the validated range, supported by linearity and hook-effect testing.
Lower variability thanks to careful handling of the biotinylated drug and the use of higher working volumes.
These attributes enabled confident interpretation of ADA data, a clear picture of the immunogenicity profile, and strong support for regulatory discussions during clinical development.
Why Syngene
Developers of RNAi therapeutics and other advanced biologics need partners who understand both immunogenicity science and regulated bioanalysis. In this project, Syngene combined experience with large-molecule ADA assays, MSD electrochemiluminescence technology, and handling of sensitive conjugated reagents within advanced bioanalytical laboratory solutions for biologics.
GxP-compliant bioanalytical laboratories, integrated with translational research and clinical development functions, provided consistent sample handling, efficient troubleshooting, and timely ADA reporting. For the sponsor, this translated into high-quality data, robust documentation, and a partner ready to support future study phases and integrated clinical development programs using the same validated method.
Bioanalytical capabilities in focus
Syngene offers advanced bioanalytical laboratory solutions for biologics, supporting large molecule bioanalysis from preclinical studies through late-stage clinical development. Dedicated GxP-compliant laboratories handle monoclonal antibodies, bispecifics, fusion proteins, RNA-based therapeutics, and other complex modalities. Teams design and execute fit-for-purpose and fully validated ligand-binding assays and cell-based assays for PK, PD, immunogenicity, neutralizing antibodies, and exploratory biomarkers on platforms such as MSD, AlphaLISA, and related immunoassay systems. Regulated bioanalysis is performed under robust quality frameworks with strong data integrity controls and 21 CFR Part 11–compliant informatics. Close integration with translational research and clinical development services enables efficient central laboratory support, streamlined global sample logistics, and rapid, decision-ready data delivery to clinical trial teams. Through this integrated model, sponsors can rely on Syngene for consistent, submission-ready bioanalytical data across the full life cycle of biologic and RNAi therapeutic programs.







