A cell-based neutralizing antibody bioassay was optimized within an advanced bioanalytical laboratory setting to control donor variability and drug interference for a clinical PD-1 checkpoint inhibitor program in oncology clinical development.
At a glance
A global sponsor developing a fully human IgG4 monoclonal antibody against programmed death receptor-1 (PD-1) required a neutralizing antibody (NAb) assay to support clinical immunogenicity assessment as part of its large molecule bioanalysis strategy. Syngene set up a cell-based bioassay using a cognate pair of mouse T and B cell lines (3A9 and LK35.2) engineered to overexpress human PD-1 and PD-L1. A bead extraction with acid dissociation (BEAD) step and immunodepletion pretreatment reduced drug interference. Targeted troubleshooting addressed high variability in normal human donor sera, leading to uniform responses across individual and pooled samples within the same run. The final AlphaLISA-based readout provided a reliable, drug-tolerant NAb method suitable for regulated bioanalysis and regulated studies.
Background
PD-1 blocking antibodies have become an important class of immuno-oncology drugs. As part of clinical development, regulators expect both binding ADA and neutralizing antibody data to understand the potential impact on efficacy and safety. For this program, the sponsor needed a sensitive, drug-tolerant NAb assay that could detect antibodies blocking the PD-1/PD-L1 interaction. The assay had to use a biologically relevant, cell-based system and be robust enough to handle normal human donor sera, which can show high intrinsic variability in clinical trial samples.
Scientific challenge
The initial bioassay used a cognate pair of mouse T and B cell lines, 3A9 and LK35.2, engineered to overexpress human PD-1 and PD-L1 so that the system mirrored the drug’s mechanism of action. However, early pre-validation work revealed two main problems. Normal individual donor sera showed high donor-to-donor variability, which made it hard to set stable cut points and compare samples across assay runs. In addition, residual therapeutic in clinical samples could interfere with the readout and hide or confuse the detection of neutralizing antibodies. These issues had to be resolved before the assay could be considered suitable for use in a GxP-regulated bioanalytical setting.
Syngene’s approach
Mechanism-reflective cell-based bioassay
Syngene refined the bioassay using the 3A9 (T cell) and LK35.2 (B cell) lines as a cognate pair. Overexpression of human PD-1 and PD-L1 ensured that the assay reflected the checkpoint pathway targeted by the IgG4 antibody. AlphaLISA technology was used as the final readout, providing a sensitive, homogeneous assay format suited for higher-throughput clinical sample testing in a neutralizing antibody functional assay.
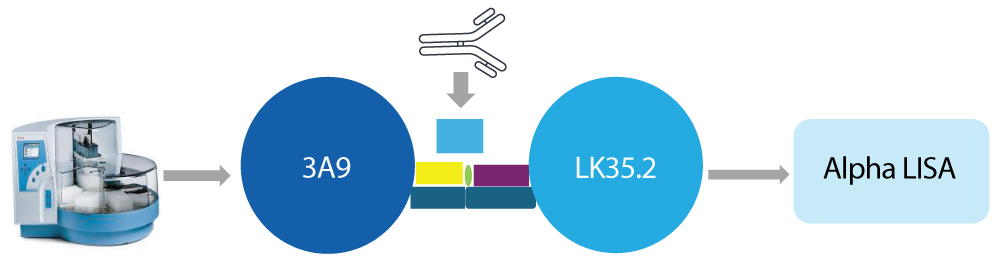
Reducing drug interference through BEAD extraction
To mitigate drug interference, Syngene incorporated a bead extraction with acid dissociation (BEAD) step. Serum samples were pretreated using immunodepletion on a Kingfisher Flex platform, followed by acid-mediated dissociation to release antibodies from circulating drugs. This pretreatment reduced the amount of free therapeutic carried into the functional readout and allowed neutralizing antibodies to be measured more accurately within the NAb bioassay.
Controlling donor variability
To control donor variability, a focused troubleshooting strategy was used for normal donor sera. Experiments compared responses from individual donor sera and pooled sera within the same assay run, which helped optimize conditions that produced more uniform results. Elution buffer from the extraction step was also spiked with drug and evaluated, along with media-alone controls, to check that neither the buffer components nor residual drug generated a nonspecific signal. Together, these experiments showed that the assay could distinguish true neutralizing activity from background effects and that external components did not interfere with the AlphaLISA readout in this bioanalytical immunogenicity assay.
Outcome and impact
The optimized NAb bioassay delivered several benefits for the PD-1 antibody program:
A biologically relevant cell-based system that mirrors the PD-1/PD-L1 interaction and supports clinical immunogenicity assessment.
Improved drug tolerance through BEAD-based extraction and immunodepletion, reducing the impact of residual therapeutic.
Demonstrated uniformity of responses across normal donor sera and serum pools within the same run, supporting reliable cut point determination and consistent data across clinical studies.
A consistent AlphaLISA readout suitable for regulated immunogenicity studies and long-term clinical sample testing within GxP-compliant bioanalytical laboratories.
Together, these elements provided the sponsor with a dependable neutralizing antibody method that could be used alongside binding ADA assays to characterize the immunogenicity profile of the PD-1 blocking antibody throughout clinical development.
Why Syngene
Sponsors developing checkpoint inhibitors and other complex biologics need partners who can translate the mechanism of action into robust functional assays. In this project, Syngene combined expertise in cell-based bioassays, neutralizing antibody method development, and drug tolerance strategies such as BEAD extraction and immunodepletion as part of its advanced bioanalytical laboratory solutions for biologics.
GxP-compliant bioanalytical laboratories, integrated sample management, and experience with donor variability allowed Syngene to troubleshoot pre-validation challenges quickly and deliver a fit-for-purpose assay for large molecule bioanalysis in clinical development. This capability helps sponsors generate high-quality NAb data, respond confidently to regulatory expectations, and maintain continuity of immunogenicity assessment across clinical phases and global oncology programs.
Bioanalytical capabilities in focus
Syngene offers advanced bioanalytical laboratory solutions for biologics, supporting large molecule bioanalysis from preclinical studies through late-stage clinical development. Dedicated GxP-compliant laboratories handle monoclonal antibodies, bispecifics, fusion proteins, RNA-based therapeutics, and other complex modalities. Teams design and execute fit-for-purpose and fully validated ligand-binding assays and cell-based assays for PK, PD, immunogenicity, neutralizing antibodies, and exploratory biomarkers on platforms such as MSD, AlphaLISA, and related immunoassay systems. Regulated bioanalysis is performed under robust quality frameworks with strong data integrity controls and 21 CFR Part 11–compliant informatics. Close integration with translational research and clinical development services enables efficient central laboratory support, streamlined global sample logistics, and rapid, decision-ready data delivery to clinical trial teams. Through this integrated model, sponsors can rely on Syngene for consistent, submission-ready bioanalytical data across the full life cycle of biologic and RNAi therapeutic programs.







