The Rising Challenges of Modern Drug Discovery
Eroom’s Law highlights a troubling trend in pharmaceutical R&D: the cost and time required to develop new drugs have been steadily increasing despite technological advances (1). As a result, drug discovery is under constant pressure. Early-stage research has become increasingly complex, fast-moving, and resource-intensive, such that most organizations cannot or choose not to manage it alone. The challenges in drug discovery are driven by multiple factors: R&D timelines can take 10-14 years from discovery to market approval (2,3,4), costs per approved drug exceed US$2.6 billion (5), and clinical success rates hover at just 14.3% (6). In the context of inefficiency, risk, and escalating investment, fragmented approaches to outsourcing add a layer of complexity that ranges from impractical to unsustainable.
From Fragmented to Integrated: Rethinking CRO Roles in Modern Drug Discovery
Western large pharma and a significant percentage of emerging pharma/biotech companies have traditionally managed externalized research efforts to Asian CROs in a manner more transactional and directive, as opposed to collaborative. Efficiency gains have, at best, been realized through the co-location of functional services. However, over the last ~5-7 years, there has been a significant shift towards viewing CROs as potential partners in innovation, in addition to the more traditional functional services provision. Hence, the recent advent of the terminology “Integrated Innovation Partner” (IIP).
Companies are increasingly leveraging IIPs for authentic integrated drug discovery to gain the benefits of leveraging external expertise while simultaneously reducing handovers to maintain momentum. In the best case, an IIP can bring a client all the way from client ideation (including target identification and validation) through to preclinical candidate nomination to clinical proof of concept – and beyond. In this way, maximum speed can be derived in the context of the flexibility and potential cost advantages of working with a qualified Asian provider.
Three main factors shape this evolution, and these factors remain the top drivers for CRO growth. First, there is sustained pressure to meet tight deliverables and timelines, demanding greater efficiency and agility in how projects are planned and executed. Second, sponsors need dependable access to specialized expertise and leading-edge technology so that programs can move from idea to outcomes without bottlenecks. Third, relentless cost pressures require models that control spend while protecting quality and reproducibility, ensuring value across the discovery lifecycle.
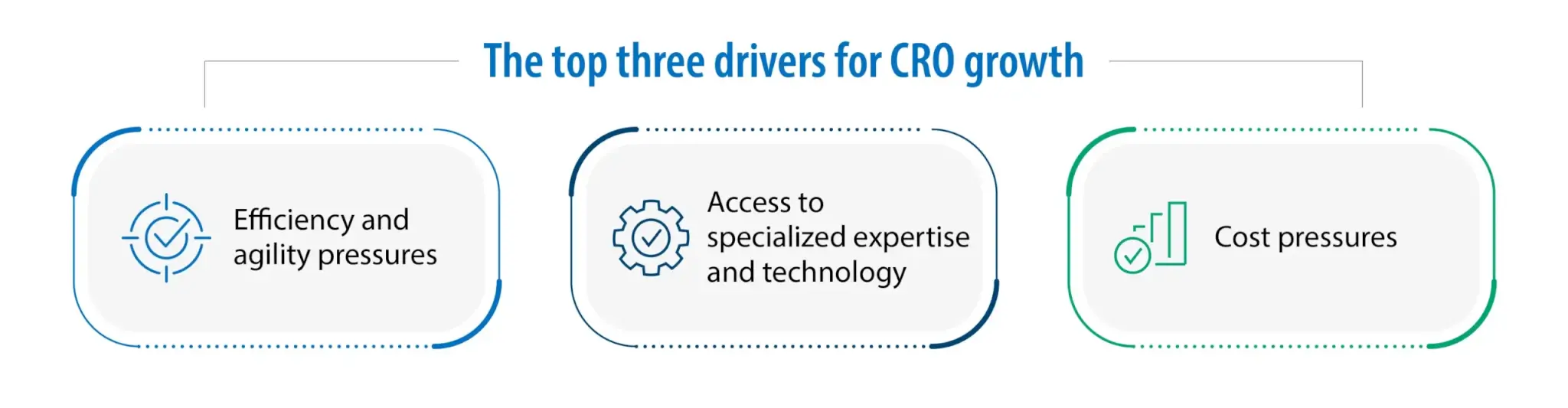
From tasks to partnerships in integrated drug discovery services
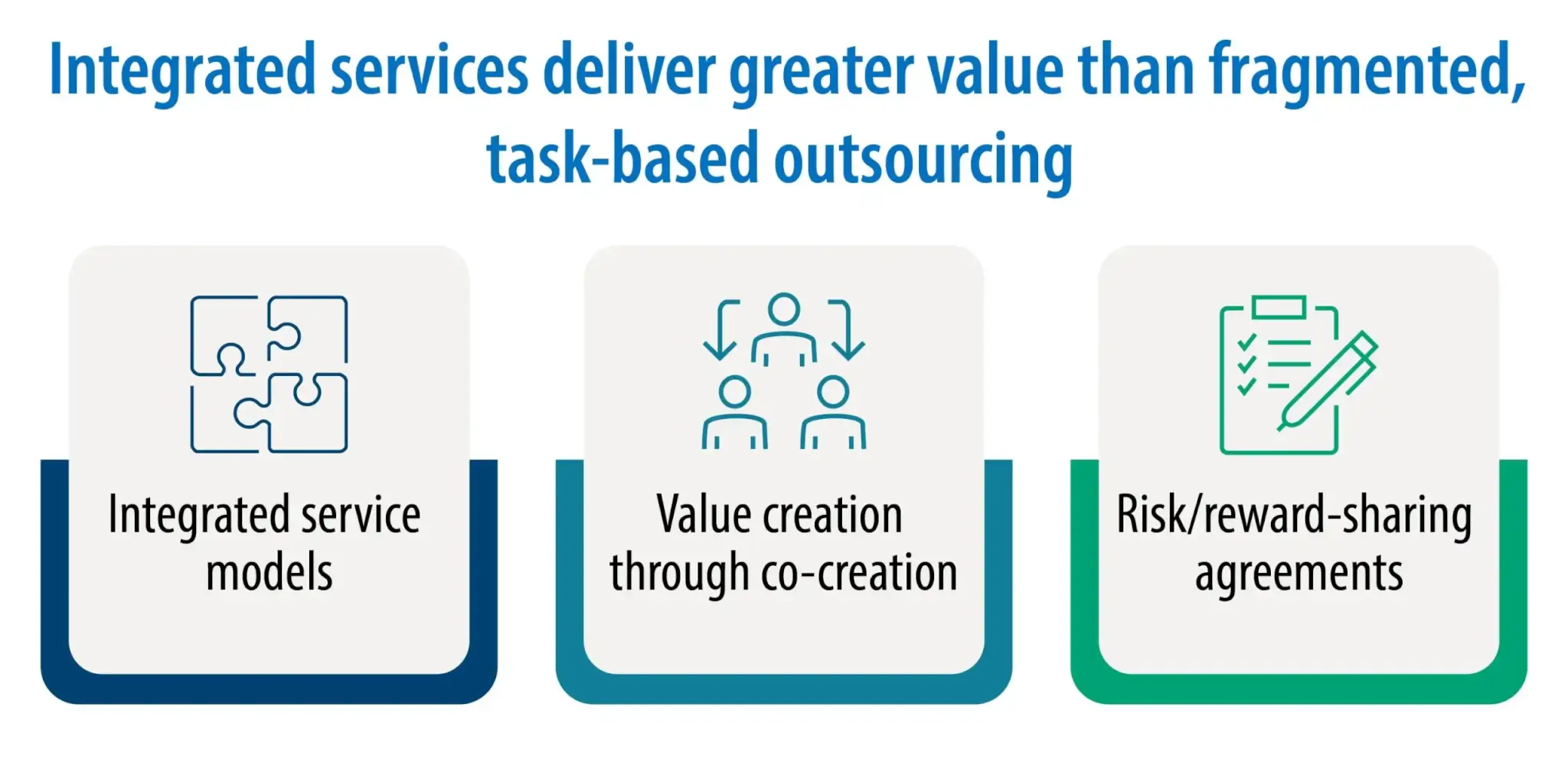
While the convergence of these drivers underscores the classic trio of speed, quality, and cost to dominate CRO selection, the field is evolving. The future belongs to CROs/IIPs that truly transition from dedicated contract service providers to broadly capable, risk-aligned, and interconnected partners in ways that redefine the industry. This shift is now evident in the movement away from fragmented, task-based outsourcing toward long-term partnerships, integrated service models, and value creation through co-creation and risk/reward-sharing agreements. Not just “integrated drug discovery” framed primarily in cost-saving terms, but the ability to bring problem-solving capability, technological innovation, transparent communication, and trust to support strategic, long-term partnerships with shared innovation goals.
Syngene SynVent™: Reimagining Integrated Drug Discovery
Many traditional task-based models still force a trade-off between cost and capability and remain constrained by legacy structures, institutional bias, and uneven access to depth and diversity of insight. Western providers bring decades of experience but at a premium that can be prohibitive. Asian providers offer cost advantages but may lack the institutional knowledge built over years in top-tier, Western pharma and biotech environments. Even when budget isn’t the issue, project teams are frequently led by a handful of senior scientists overseeing junior staff—limiting the depth and diversity of insight.
Institutional Knowledge Without Institutional Bias
With SynVent, there is a better way: combine institutional knowledge without institutional bias with agile, highly cost-effective execution. This ensures your project benefits from rich experience across many of the world’s leading pharmaceutical and biotech companies—without being confined by the biases of any single organization.
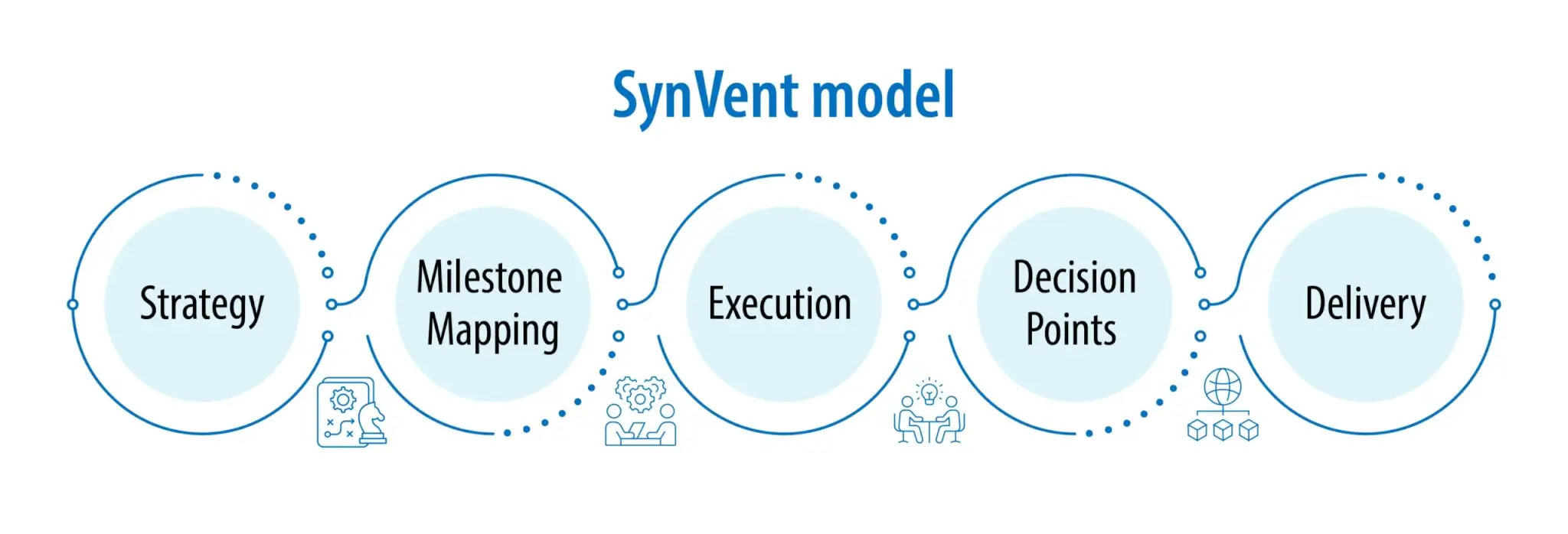
From strategy formulation through execution, SynVent leaders remain actively involved throughout the lifecycle of the project—participating with clients to define the research plan, map milestones, advise on resources, and guide decisions. SynVent project teams are optimized to ensure seamless collaboration, scientific rigour, and strategic clarity, leveraging the deep expertise typically found in Western institutions with the capabilities at scale within the largest CRDMO in India, which brings 32 years of experience to client projects.
What Drives SynVent’s Approach
Talent is pivotal to accelerating innovation
The SynVent leadership team is composed entirely of seasoned drug hunters with decades of experience across many of the world’s leading pharmaceutical and biotech companies and understands what it takes to propel molecules from concept to clinic. This breadth ensures institutional knowledge—without being constrained by any single organization’s way of working.
Execution guidance
From strategy to delivery, leaders partner closely with the client—co-creating strategies, aligning on data-driven decision points, and ensuring clarity on milestones and resources. SynVent does not just manage or deliver services—it drives innovation in collaboration with and on behalf of clients.
From Transaction to Transformation
The industry’s direction is clear: move beyond task-based outsourcing toward integrated discovery where capability, speed, quality, cost, and trust reinforce each other. Syngene SynVent™ embodies this direction: not just another CRO, but a strategic partner committed to transforming how drug discovery gets done—through long-term partnerships, co-creation, and shared accountability.
Let’s start a conversation.
Are you exploring new models for drug discovery? Curious how Synvent could accelerate your pipeline? Write to us bdc@syngeneintl.com