Your Integrated Bioanalytical Partner for Translational and Clinical Success

Syngene, with its integrated bioanalytical capabilities across small and large molecules, delivers accurate, regulatory‑compliant data to support programs from discovery through clinical development.
Built-for-purpose Biomarker Solutions Enhancing Speed, Precision, and Confidence

Syngene, with its comprehensive biomarker services spanning discovery, validation, and context‑of‑use assays, enables early decision‑making, patient stratification, and precision‑led drug development.
Next-Gen Modalities, Expertly Enabled

Syngene’s integrated capabilities in Oligonucleotides, Peptides, & Conjugates. From discovery to GMP, we partner with you to accelerate novel modalities.
CPHI Japan 2026 Takeaways: Why supply chain diversification is redefining CDMO trust in Japan

Explore how supply chain diversification is changing CDMO selection in Japan, from technical depth to global operating models. Read more.
Integrated gene to GMP services with mAb accelerator
Integrated gene to GMP services with mAb accelerator – Get a 4‑month head start with Syngene’s mAb Accelerator Program, designed to fast‑track biopharma success through early investment, risk sharing, and flexible commercial models.
Decoding the FDA’s AMT program through process analytical technology

Learn how process analytical technology enables advanced manufacturing technologies under the FDA AMT program and transforms pharma production. Explore the insights today.
Targeted Protein Degradation and Novel Modalities: Getting on the Frontline
Publication: Fierce Pharma
Gene editing in 2025: from CRISPR to next generation platforms
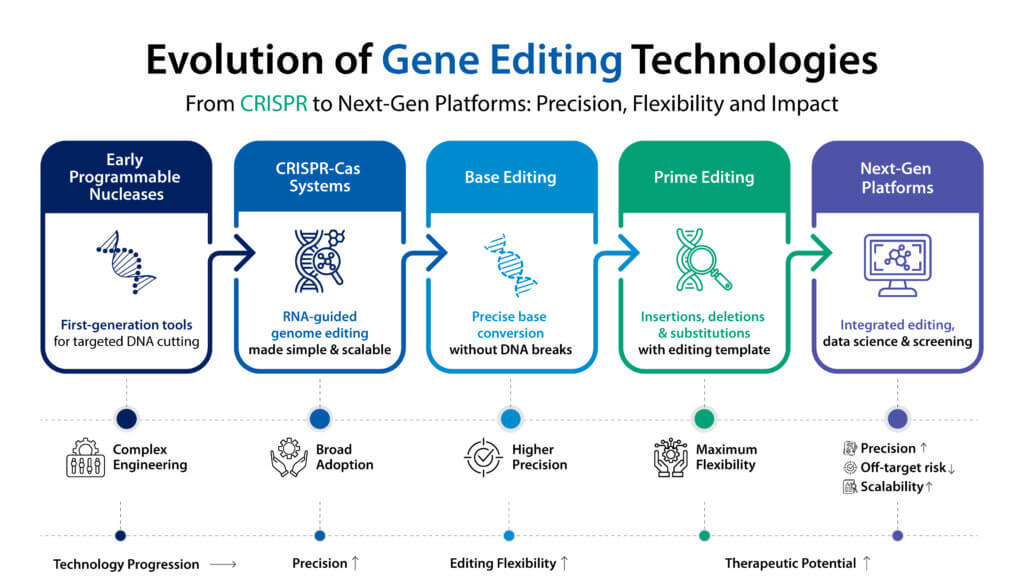
Explore how gene editing is evolving beyond CRISPR with base editing and prime editing technologies shaping next generation therapeutics.
AI Drug Discovery: The Future of Small Molecule Design
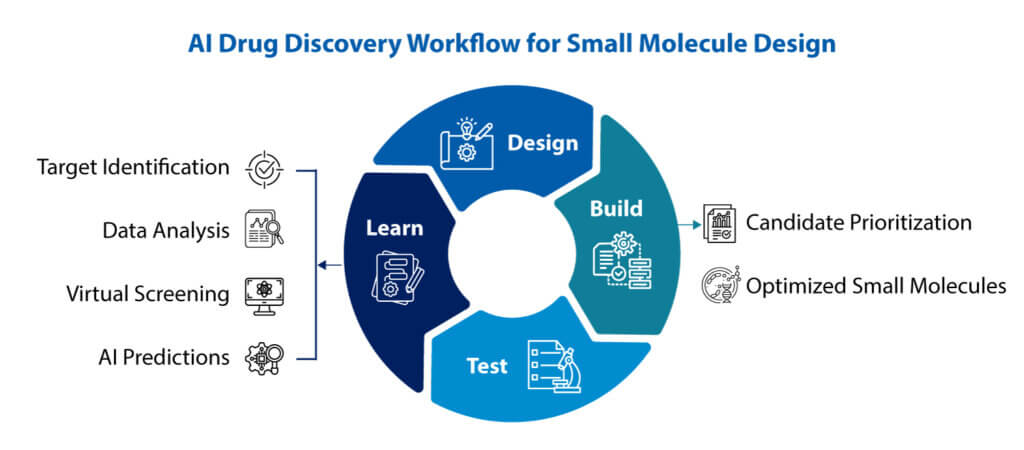
Discover how AI drug discovery is reshaping small molecule design through faster learning cycles, better prediction, and integrated decisions.
Accelerating Your Success in Antibody Discovery and Engineering

Syngene provides multi-platform antibody discovery and optimization services to drive faster progression from hits to lead candidates.






