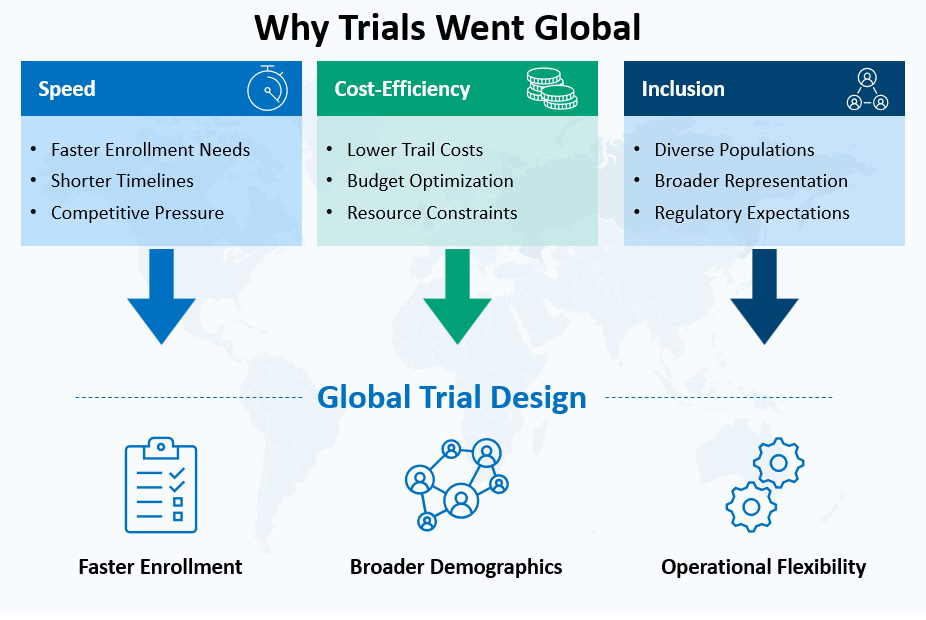
Clinical trials sit at the heart of modern drug development. They are the bridge between promising science and real-world patient benefit. Yet the way trials are planned and executed has changed sharply in the last decade. Sponsors are dealing with complex biologics, precision medicines, combination therapies, and indications where standard endpoints are evolving. At the same time, development timelines are under pressure from competitive markets, shorter product lifecycles, larger investments and higher investor expectations to achieve faster, more cost-effective, and higher-quality trial execution and treatment options.
In this environment, global clinical trials have become essential. They help sponsors recruit faster, generate more representative evidence, and manage costs without losing scientific rigor. Many therapies now target populations that are spread across multiple regions. Even when a disease burden looks concentrated in one geography, regulators and payers often expect evidence that works across demographic groups, healthcare systems, and patterns of care.
There is also a practical reality. Investigational site and patient availability in traditional trial hubs has tightened. Trial density in North America and parts of Western Europe has increased, creating competition for the same investigators and patient pools. Start-up timelines can stretch due to and administrative bottlenecks including contracting, capacity constraints at sites (infrastructure and qualified and experienced research staff) , and slow recruitment in some indications. This is pushing sponsors to think globally from the beginning rather than expanding later as a rescue strategy.
Global trials are also tied to a deeper scientific requirement: diversity. Drug response is shaped by genetics, co-morbidities, diet, environmental exposure, and healthcare access. Evidence built on narrow patient subsets can lead to surprises in safety, dosing, or effectiveness when the therapy reaches wider populations. Inclusive research is therefore becoming a quality issue, not just an ethics checkbox.
The result is a new clinical development playbook. It relies on strategic partnerships, integrated execution models, and thoughtful expansion into emerging markets such as India, Southeast Asia, and Latin America. These shifts are changing how trials are designed, how sites are selected, and how regulators evaluate evidence.
Strategic global partnerships: driving faster, smarter trials
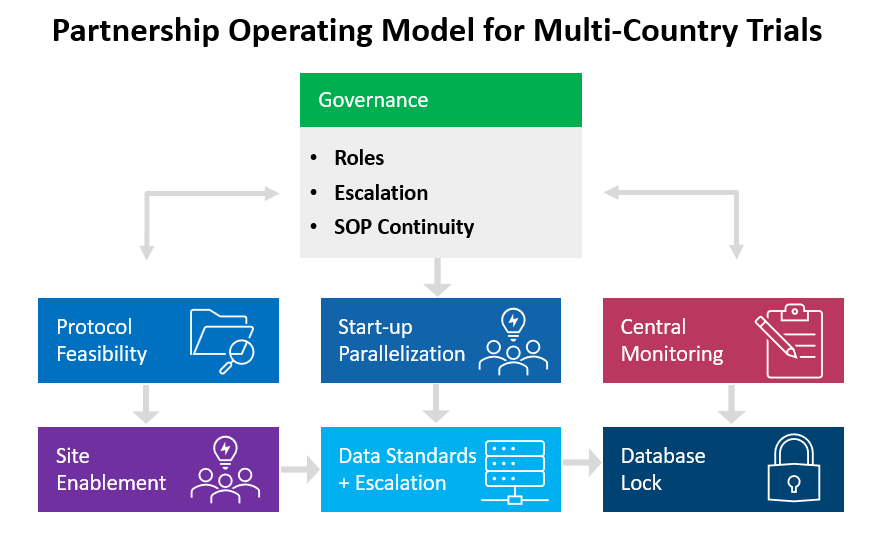
Global trials are difficult to run when execution is fragmented. Many delays come from misalignment across countries, such as different interpretations of the protocol, inconsistent investigator training, and uneven data practices. Strategic partnerships are a way to reduce this friction. When sponsors build structured collaborations across geographies, they can create a repeatable system for multi-country studies rather than reinventing processes for each program.
Partnerships are taking multiple forms. Some sponsors create preferred provider models with CROs that operate across regions. Others use integrated networks that connect central labs, imaging vendors, logistics partners, and specialist recruitment providers. Increasingly, sponsors also collaborate with hospital networks and academic groups that have multi-site reach within a country or across neighbouring countries. These models matter because global execution is less about a single vendor and more about how the ecosystem works together.
A well-designed partnership can shorten timelines in several ways. First, feasibility becomes more realistic. If partners understand regional patient pathways, standard of care, and referral patterns, recruitment assumptions improve and site selection becomes sharper. Second, a start-up can run in parallel. With shared templates, pre-agreed contracting approaches, and established quality expectations, ethics submissions, site initiation, and vendor onboarding do not have to be sequential or improvisational. Third, operational learning carries forward. Teams do not lose time repeating the same mistakes because there is continuity of tools, SOPs, and governance practices.
CROs are often at the center of these partnerships, but the value has shifted. Execution capability is still important, yet mature sponsors expect more than staffing and project management. They want regional regulatory intelligence, proactive risk identification, and quality-by-design thinking. For example, a strong partner can flag early that a proposed endpoint requires local validation, that a diagnostic is not widely available in certain regions, or that patient travel burden is likely to increase dropout risk.
The partnership conversation also extends to data. Multi-country studies depend on consistent data capture, query handling, and audit readiness. This becomes easier when sponsors and partners align on core data standards, define escalation pathways for data issues, and ensure that systems are interoperable. When these elements are missing, sponsors often face painful “data clean-up phases” late in the trial. That clean-up can delay database lock and create stress during regulatory submission.
The strongest partnership models share some common traits:
- Governance that is simple and decisive. Clear roles, clear escalation, and clear ownership of actions.
- Consistent training and oversight. Investigators in different regions should receive the same protocol intent, not just the same slide deck.
- Central quality systems with local sensitivity. Standardization should not ignore local operating realities, but it should prevent variability that harms trial integrity.
- Early feasibility realism. Over-optimistic enrolment forecasts remain a major cause of global trial delays.
When these foundations are in place, global trials become more predictable. Predictability is often more valuable than a small reduction in unit cost because it protects timelines and reduces the need for mid-study redesign.
Emerging markets: unlocking cost-effective and high-quality trials
Emerging markets have become strategic contributors to global clinical trials, and the reasons go beyond lower per-patient costs. Sponsors are looking for faster recruitment, broader patient representation, and sites with strong clinical expertise. India, Southeast Asia, and Latin America offer a combination of these advantages, though each region has its own profile.
Why sponsors are looking at India, Southeast Asia, and Latin America
A major driver is patient availability. Many diseases have high prevalence in these regions, including diabetes, cardiovascular disease, respiratory disorders, infectious diseases, and certain cancers. Large urban hospitals often see significant patient volumes, which can speed up recruitment when trials are well matched to local care pathways.
These regions also offer an improved clinical infrastructure. Large hospitals in India and several Southeast Asian countries now have advanced imaging, specialized oncology services, and experienced research teams. In Latin America, countries such as Brazil, Mexico, Argentina, and Colombia have built strong capabilities for Phase II and Phase III trials, especially in therapeutic areas where they have established investigator communities and potential investigational sites (Centres of excellence).
Skilled workforce availability is another key factor. Investigator quality is shaped by clinical training, research exposure, and site-level operations. Many emerging markets have a growing pool of clinicians trained in global standards of care and experienced with international protocols. Alongside investigators, there is also a growing professional ecosystem of clinical research coordinators, data managers, pharmacovigilance staff, and quality personnel.
Cost advantages still matter, but in a more nuanced way. Lower operational costs can support larger sample sizes, additional sub-studies, or broader geographic coverage. More importantly, faster recruitment can reduce the hidden costs of extended timelines. A study that completes enrolment six months earlier can save substantial program costs even if per-patient costs are only moderately lower.
Quality and compliance expectations
The main question sponsors ask is whether speed and cost come at the expense of quality. In serious programs, the answer must be no. Emerging markets that succeed in global trials do so because they can meet international GCP expectations and support audit readiness. Strong sites demonstrate consistent documentation practices, stable staff training, reliable source data verification, and robust handling of informed consent. Sponsors also look for sites that can manage protocol complexity, including biomarker-driven recruitment, imaging-based endpoints, and intensive safety monitoring.
The reality is that quality varies by site, not just by country. Mature sponsors avoid treating a region as uniformly strong or weak. They invest in careful site qualification, study-specific training, and ongoing oversight. When done well, emerging market sites can deliver data that stands up to scrutiny and supports global submissions.
Practical considerations that shape performance
Several operational factors influence whether emerging markets deliver their promise:
- Start-up timelines depend on local contracting norms, language requirements, and document flows between sponsor, CRO, and sites.
- Patient retention depends on travel burden, visit schedules, and local support systems.
- Diagnostics and standard of care influence eligibility. If the protocol requires tests that are not routinely available, recruitment slows.
- Supply chain and cold-chain logistics matter, especially for biologics, cell therapies, and temperature-sensitive products.
- Data connectivity and system reliability affect how smoothly electronic systems run at sites.
Sponsors who plan for these realities do better than those who assume a region will automatically deliver faster outcomes.
Diversity in clinical trials: why global collaboration matters
Diversity in clinical research is now seen as central to scientific validity. A therapy intended for broad patient use should be tested in populations that reflect real-world diversity. This includes ethnicity, age groups, sex, co-morbidities, body weight ranges, and socio-economic contexts. Without this, trial results can be incomplete, and risk assessments can become fragile.
There are several reasons diversity matters from a clinical perspective:
- Genetic differences can influence drug metabolism and immune response.
- Baseline disease severity and co-morbidities vary across populations.
- Diet, lifestyle, and environmental exposure can affect outcomes.
- Patterns of care differ, which can change adherence and follow-up.
A global trial design supports diversity by creating access to multiple demographic pools. For example, recruiting across Asia, Latin America, and Europe can increase representation in a way that is hard to achieve through local recruitment alone. It also helps in identifying where a disease is more prevalent in specific populations.
Diversity is also tied to regulatory expectations and public trust. Regulators want confidence that trial evidence can translate to the populations who will use the medicine. Patient groups and advocacy communities increasingly ask who was included in trials and whether exclusions were justified. Sponsors are being pushed to show intentional planning rather than accidental diversity.
However, diversity does not happen automatically. Sponsors need to plan it with the same seriousness as sample size and power calculations. This means setting recruitment targets, selecting sites with the right patient access, reducing participation barriers, and using community engagement where appropriate. It also means designing protocols that do not exclude common real-world patients without strong scientific reasons. Overly restrictive criteria remain a frequent barrier to inclusion.
Global collaboration can also support ethical inclusion when done responsibly. Local relevance matters. Trials should address diseases and needs that exist in the participating regions. Informed consent must be culturally and linguistically appropriate. Post-trial access and continuity of care should be planned, particularly for serious conditions. A global trial that respects local context is more likely to deliver both strong data and strong relationships with sites and communities.
India’s evolving regulatory landscape: a catalyst for global trials
India is often discussed as a key emerging market for clinical research, and its role is closely linked to regulatory evolution. Over time, India has introduced reforms aimed at improving clarity, strengthening oversight, and making processes more predictable for sponsors and investigators.
Several elements have contributed to improved feasibility. Defined review timelines reduce uncertainty during start-up. Greater structure around ethics committees strengthens oversight and standardizes expectations. Increased use of digital submission pathways supports transparency and reduces delays from manual workflows. These changes matter because predictability is often what sponsors seek most when choosing a country for global trials.
Regulatory alignment with international norms also builds sponsor confidence. When local requirements are consistent with global expectations, protocol design becomes simpler and operational teams face fewer country-specific deviations. This supports smoother multi-country execution and reduces the risk of last-minute changes that disturb timelines.
India also brings specific strengths. Large patient volumes in major hospitals can support recruitment, especially for high-burden conditions. There is a strong base of specialist physicians in urban centers. Many leading hospitals have built dedicated research teams and infrastructure. The ecosystem is also supported by a growing group of professionals in clinical operations, data management, quality, and pharmacovigilance.
At the same time, sponsors must approach India with realistic planning. Site performance can vary widely, so careful selection is essential. Competition for top-tier sites exists. Patient retention and follow-up need attention, especially for trials with frequent visits. Protocols should be designed with the local standard of care in mind to avoid feasibility issues.
When these aspects are addressed, India can contribute strongly to global trials, both in recruitment speed and in diversity contribution.
Looking ahead: the next phase of global clinical research
The next five years are likely to bring further changes in how global trials are conducted. Three trends stand out: decentralized elements, technology-enabled oversight, and gradual regulatory convergence.
Decentralized and hybrid trials
Decentralized trials are expanding, but full decentralization is not realistic for many studies, especially in oncology and complex biologics. Hybrid models are more common. They blend site-based procedures with remote components such as tele-visits, home nursing, local lab draws, and eConsent. In global trials, hybrid approaches can help reduce patient burden, improve retention, and support recruitment in regions where travel to sites is difficult.
However, decentralized elements need careful planning. Data integrity, privacy, and oversight requirements vary by region. Home health providers must be qualified. Remote assessments must be validated and consistent. Sponsors will likely become more selective, choosing decentralized tools that genuinely improve outcomes rather than adding complexity.
Tech-driven monitoring and quality oversight
Risk-based monitoring has already grown, and the next phase will be more data-driven. Central monitoring can detect unusual patterns, inconsistent reporting, and safety signals earlier. This supports global studies where on-site monitoring for every visit is not feasible. Remote access to source data, where permitted, can also improve oversight.
Digital tools can help, but they can also create new failure points if systems are fragmented. Sponsors who build clear data governance and interoperability across platforms will benefit. Those who stack multiple tools without integration may face data reconciliation challenges and training fatigue at sites.
Harmonized regulations and reliance pathways
Regulatory harmonization is a gradual process, yet there is steady movement in that direction. Agencies share learnings on trial oversight, good clinical practice expectations, and data standards. Reliance pathways and mutual recognition approaches can reduce duplication in reviews, though these are not universal and vary by region.
For sponsors, the practical implication is that global trial design may become more standardized, but local nuance will remain. Protocols will still need country-specific tailoring in areas such as consent language, data privacy, and operational logistics.
What sponsors should do now
Sponsors can prepare for this future by focusing on a few core actions:
- Build partnership models that scale across programs rather than selecting vendors program by program.
- Invest in feasibility grounded in real-world patient pathways and site capacity.
- Treat diversity as a design parameter, not an afterthought.
- Strengthen data governance early, including standards, audit trails, and system interoperability.
- Use technology where it reduces burden and improves oversight, not where it adds complexity.
Organizations that excel in these areas will likely experience fewer late-stage surprises, smoother submissions, and greater confidence in their evidence packages
Conclusion
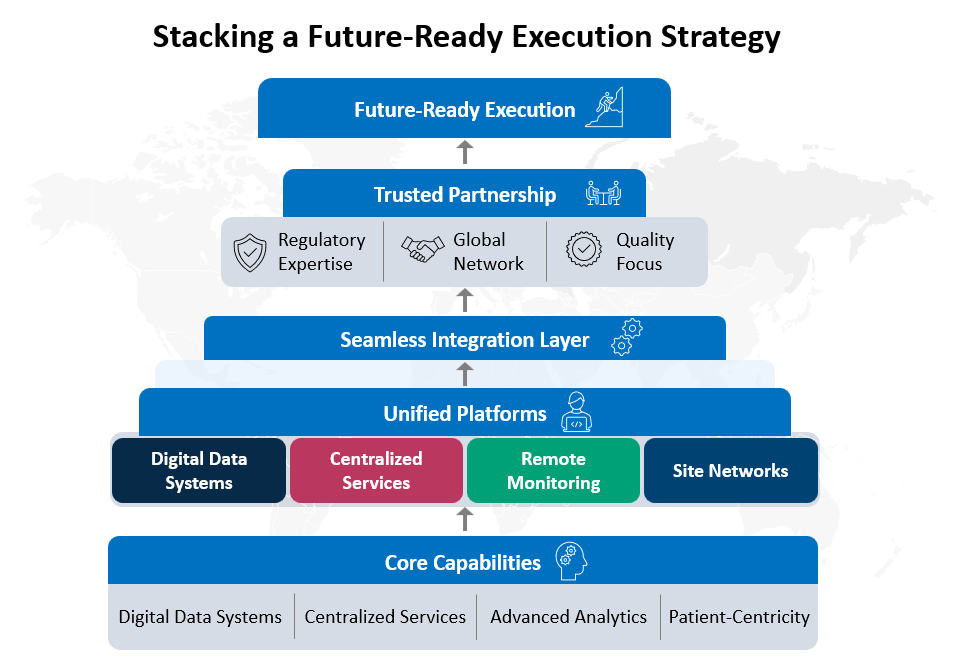
Global clinical trials are being reshaped by strategic partnerships and the expanding role of emerging markets. Sponsors are balancing speed, cost-efficiency, and data quality while responding to stronger expectations for diversity and inclusion. Regions such as India, Southeast Asia, and Latin America are no longer secondary options. They are becoming central to how sponsors deliver timely and globally relevant evidence.
The shift is not automatic. It depends on how well sponsors design partnerships, manage operational complexity, and align regulatory and data strategies across countries. As decentralized elements and technology-enabled oversight become more common, the gap will widen between programs that are thoughtfully integrated and those that are stitched together midstream.
Clinical research is moving toward a model where global collaboration is the default. Sponsors who treat this as a strategic capability, rather than a logistical challenge, will be better positioned to run inclusive trials that stand up scientifically, ethically, and regulatorily.
References
Central Drugs Standard Control Organization. (2019). The New Drugs and Clinical Trials Rules, 2019 (G.S.R. 227(E), March 19, 2019). Ministry of Health and Family Welfare, Government of India.
Central Drugs Standard Control Organization. (n.d.). New Drugs and Clinical Trials Rules, 2019. Ministry of Health and Family Welfare, Government of India.
European Medicines Agency. (n.d.). ICH E6 good clinical practice: Scientific guideline.
International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. (2025). ICH E6(R3): Guideline for good clinical practice (Step 4).
U.S. Food and Drug Administration. (2020). Enhancing the diversity of clinical trial populations: Eligibility criteria, enrollment practices, and trial designs. Guidance for industry.
U.S. Food and Drug Administration. (2023). Diversity action plans to improve enrollment of participants from underrepresented populations in clinical studies. Draft guidance for industry.
U.S. Food and Drug Administration. (2023). Conducting clinical trials with decentralized elements. Guidance for industry, investigators, and other interested parties.
World Health Organization. (2022). Global clinical trials forum: Improving diversity and access.
World Health Organization. (2024). Guidance for best practices in clinical trials.






