Neutralizing antibody assay for a fully human PD-1 blocking monoclonal antibody
Learn how Syngene’s PD-1 neutralizing antibody bioassay reduced donor variability and drug interference for immunogenicity assessment. Read the full case study today.
Reimagining CROs: Integrated Innovation Partnerships as the Future of Drug Discovery

Eroom’s Law highlights a troubling trend in pharmaceutical R&D: the cost and time required to develop new drugs have been steadily increasing despite technological advances (1). As a result, drug discovery is under constant pressure.
Immunogenicity assay for an RNAi therapeutic (small interfering RNA) on the MSD platform

Syngene’s MSD-based ADA assay enabled sensitive, drug-tolerant immunogenicity assessment for a clinical siRNA therapeutic in regulated studies. Learn more.
Improving Supply Chain Resilience: HPAPI Containment And Batch Reliability In Global Pharma Manufacturing

Global pharmaceutical supply chains are under sustained pressure from rising regulatory expectations, complex molecules, and frequent disruptions across geographies. For manufacturers handling highly potent active pharmaceutical ingredients (HPAPIs), the challenge is more pronounced.
Forging the Future: Why Strategic Outsourcing in Biologics is No Longer Optional, It’s Essential for Success
Publication: Fierce Biotechnology
Pharmacokinetics-focused bioanalysis for a half-life extended bispecific antibody in oncology
See how Syngene used MSD platform pharmacokinetics and immunogenicity assays to de-risk a half-life extended bispecific antibody in oncology. Explore the case study.
BIOSECURE Act 2025 update: what it means for biopharma outsourcing and what to do now
Publication: Pharmaceutical Technology Focus
Cell line development: faster path to a stable master cell bank
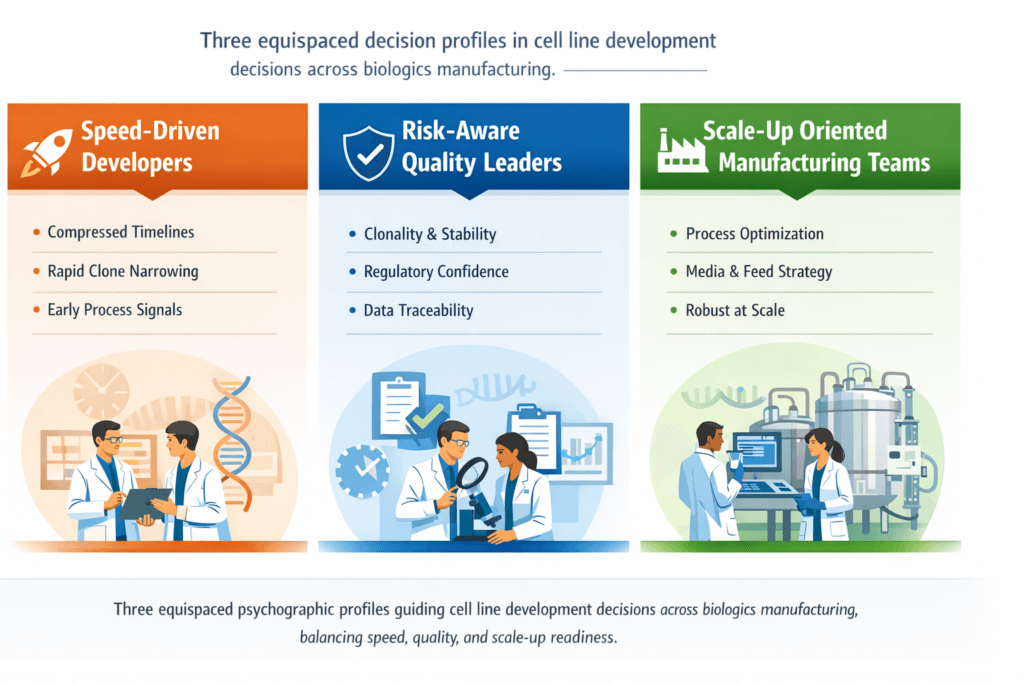
Cell line development is a critical early step in biologics manufacturing. For monoclonal antibodies and other therapeutic proteins, the quality of the master cell bank determines not only expression levels but also long-term process stability, regulatory confidence, and downstream scalability.
Redefining What’s Possible in Low-Temperature Chemistry

Read how Syngene delivers ultra‑low‑temperature chemistry down to –90 °C with precision, safety, and full GMP scalability. Explore how our cryogenic infrastructure and expertise in sensitive transformations help de‑risk development and open access to complex, otherwise inaccessible chemistries. Discover how advanced thermal control and integrated manufacturing accelerate your most challenging programs.
Partnering for Precision in Small Molecule Drug Substance Development & Manufacturing

Read how Syngene accelerates small‑molecule development with integrated discovery‑to‑commercial capabilities and globally compliant GMP facilities. Explore how advanced process chemistry, analytical depth, and seamless tech transfer de‑risk scale‑up and strengthen manufacturing reliability.






